Разделы презентаций
- Разное
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Математика
- Медицина
- Менеджмент
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, картинки для презентаций
- Экология
- Экономика
- Юриспруденция
Deinococcus radiodurans
Содержание
- 1. Deinococcus radiodurans
- 2. Content PHYSIOLOGICAL STUDIES 1. Nutritional studies 2.
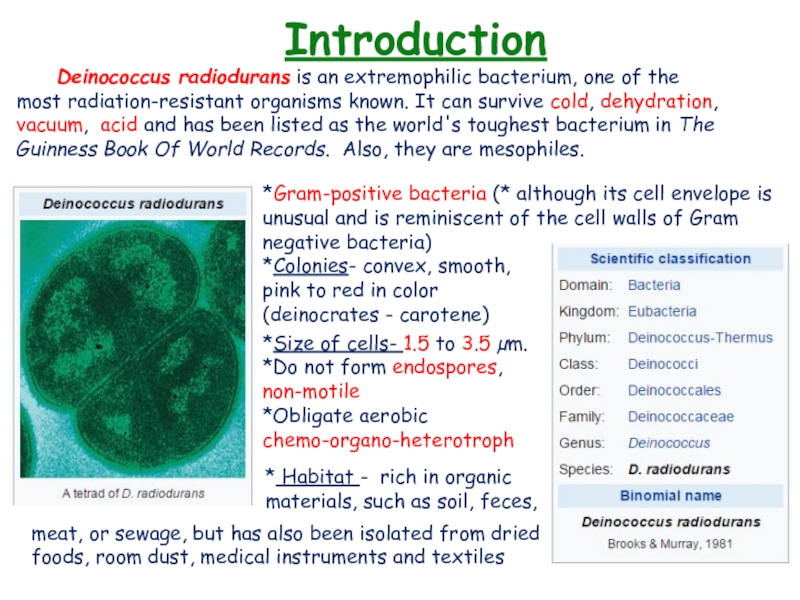
- 3. Introduction Deinococcus radiodurans is an extremophilic bacterium, one of the most radiation-resistant organisms
- 4. 1. Nutritional studies: biochemical characteristics(+++)-strong, (++)-moderate, (+)- weak, (-)-negative
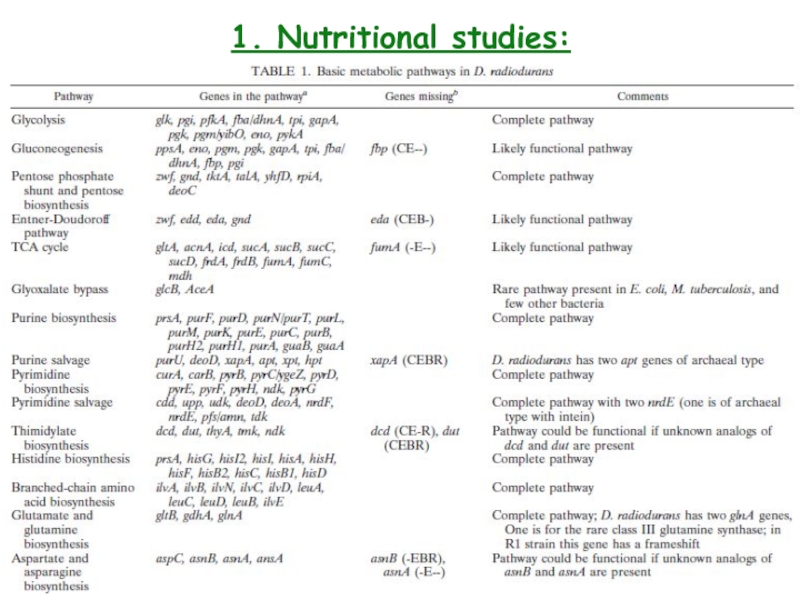
- 5. 1. Nutritional studies:
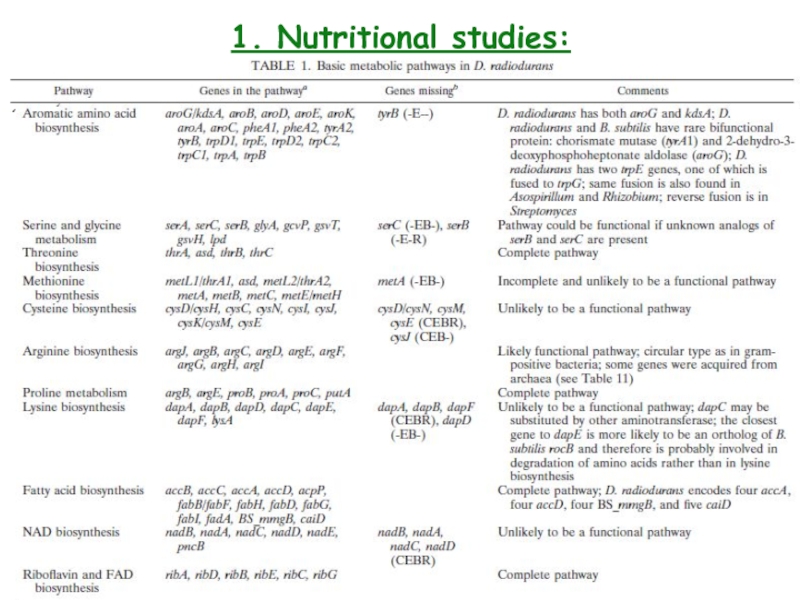
- 6. 1. Nutritional studies:
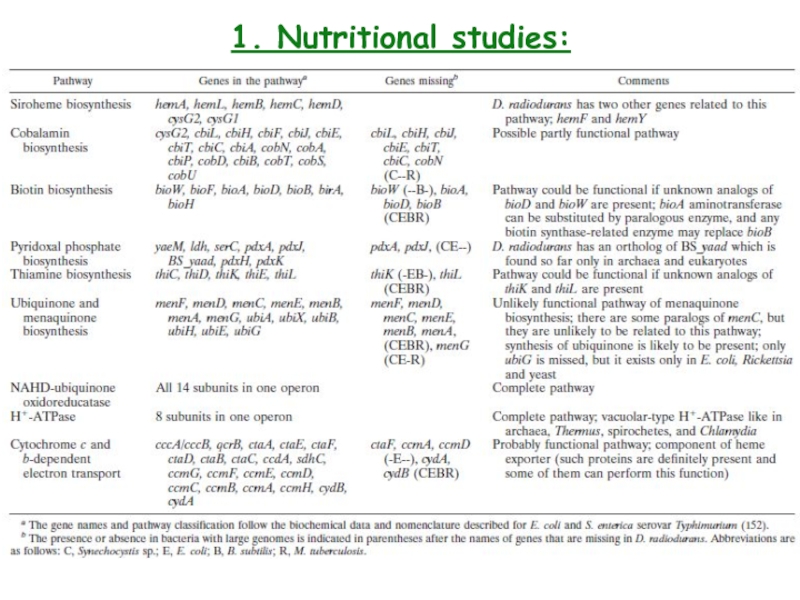
- 7. 1. Nutritional studies:
- 8. 1. Nutritional studies: nutritional medium
- 9. 1. Nutritional studies: nutritional medium
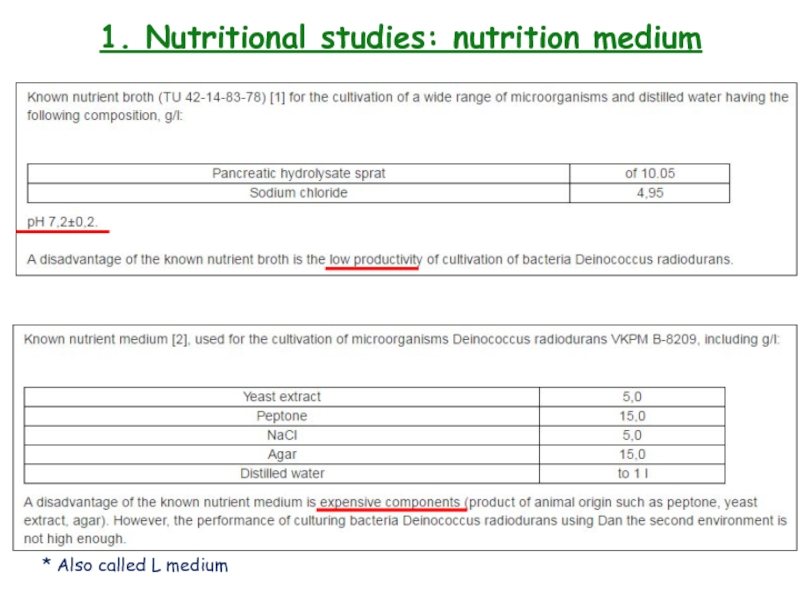
- 10. 1. Nutritional studies: nutrition medium* Also called L medium
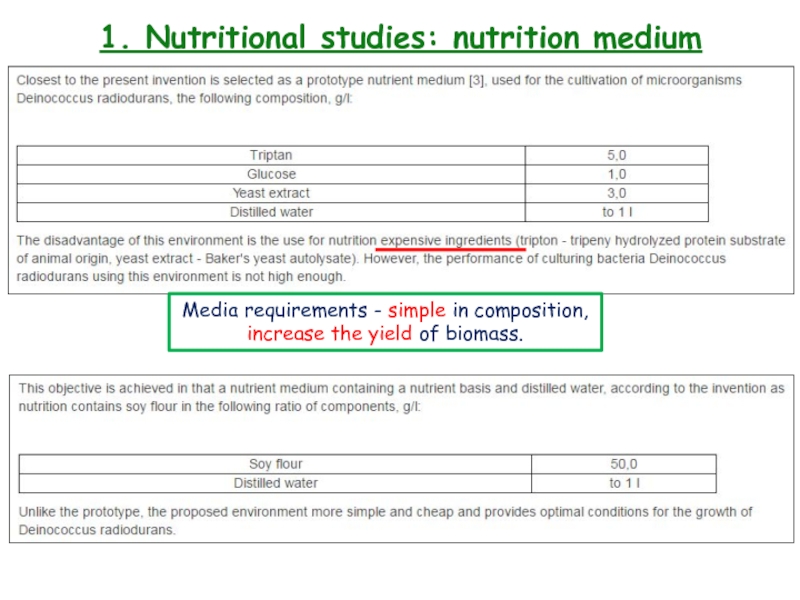
- 11. 1. Nutritional studies: nutrition mediumMedia requirements - simple in composition, increase the yield of biomass.
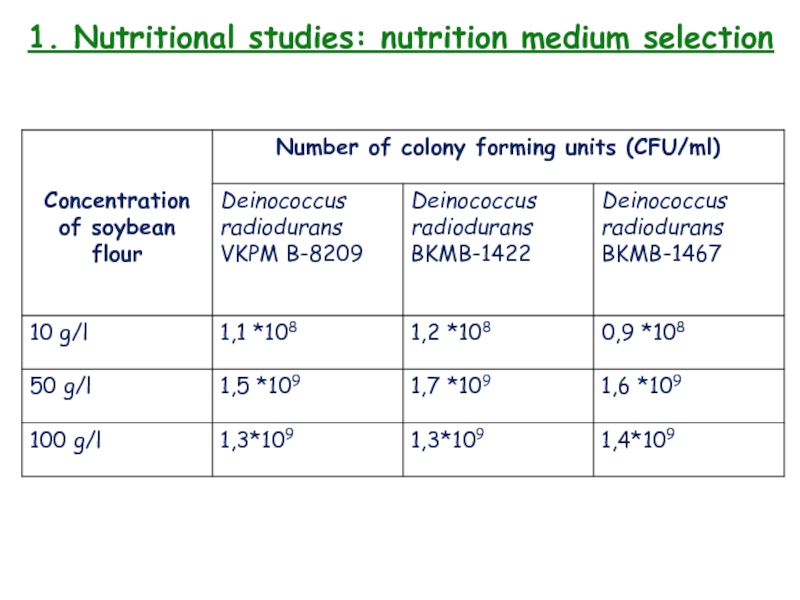
- 12. 1. Nutritional studies: nutrition medium selection3 strains
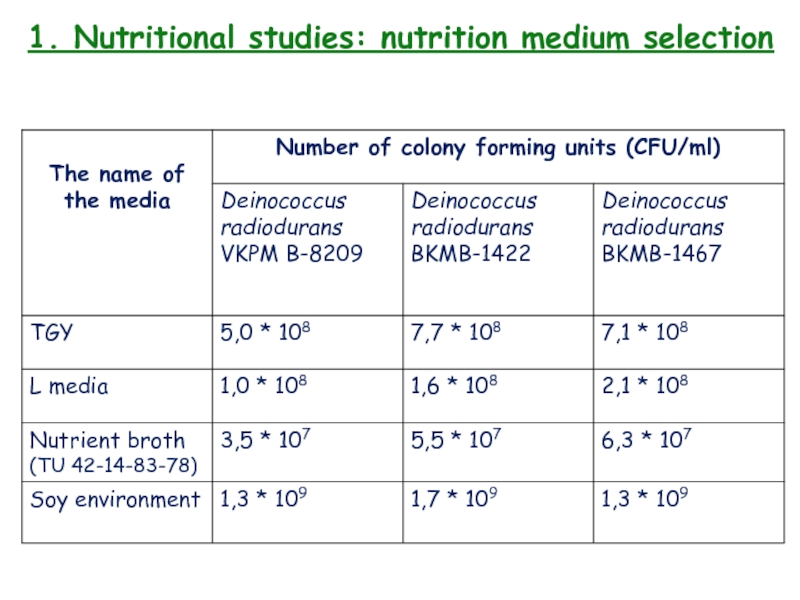
- 13. 1. Nutritional studies: nutrition medium selection
- 14. 1. Nutritional studies: nutrition medium selection
- 15. Cultivation -> Solid state/submergedBioreactor design ->Airlift bioreactor
- 16. 2. Genomic approaches The genome of D. radiodurans consists
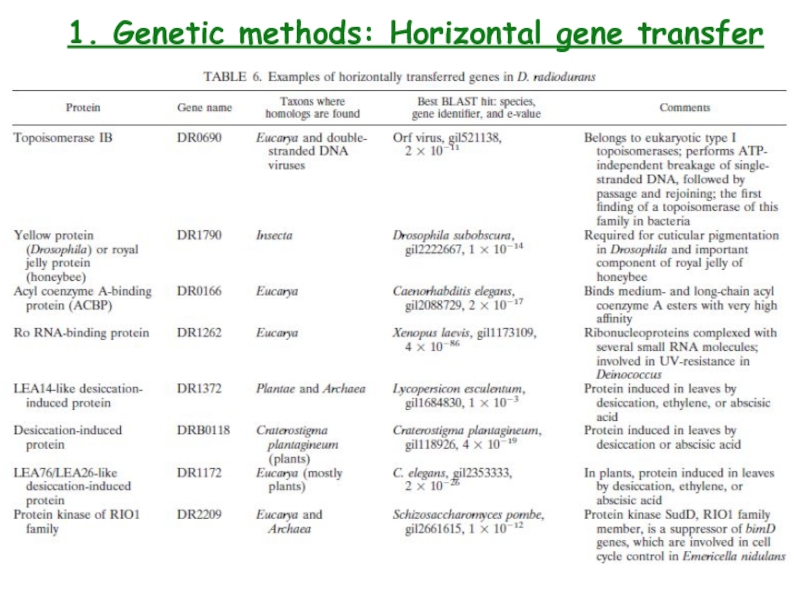
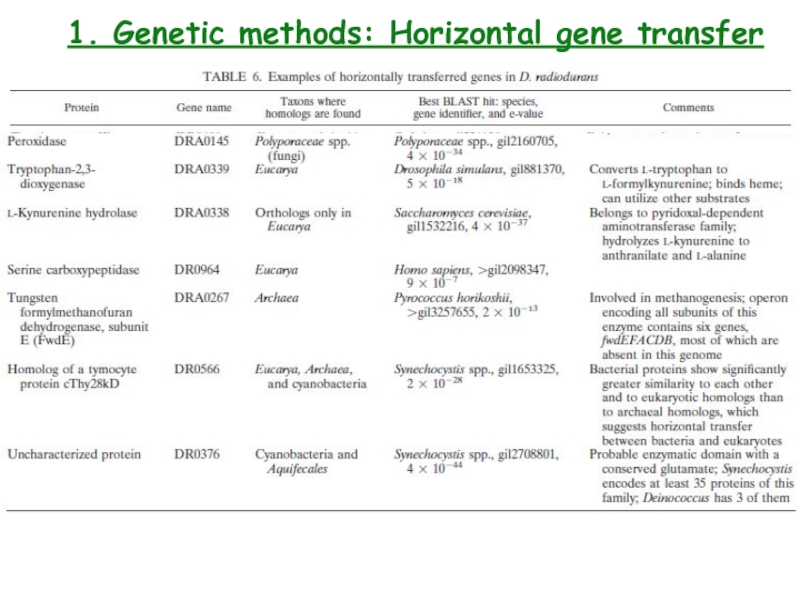
- 17. 1. Genetic methods: Horizontal gene transfer
- 18. 1. Genetic methods: Horizontal gene transfer
- 19. 1. Genetic methods: Transformation Engineering Deinococcus radiodurans for
- 20. 1. Genetic methods: Transformation pMD727 was transformed
- 21. 1. Genetic methods: Transformation pMD731 was transformed
- 22. 1. Genetic methods: Transformation Final objective: to
- 23. 1. Genetic methods: Transformation Final objective: to
- 24. 1. Genetic methods: mobile elements Inteins. Two inteins,
- 25. 1. Genetic methods: mobile elements Overall, 52 IS
- 26. 1. Genetic methods: mobile elementsAlso, it was
- 27. 1. Genetic methods: Gene inactivationInactivation of Proteins
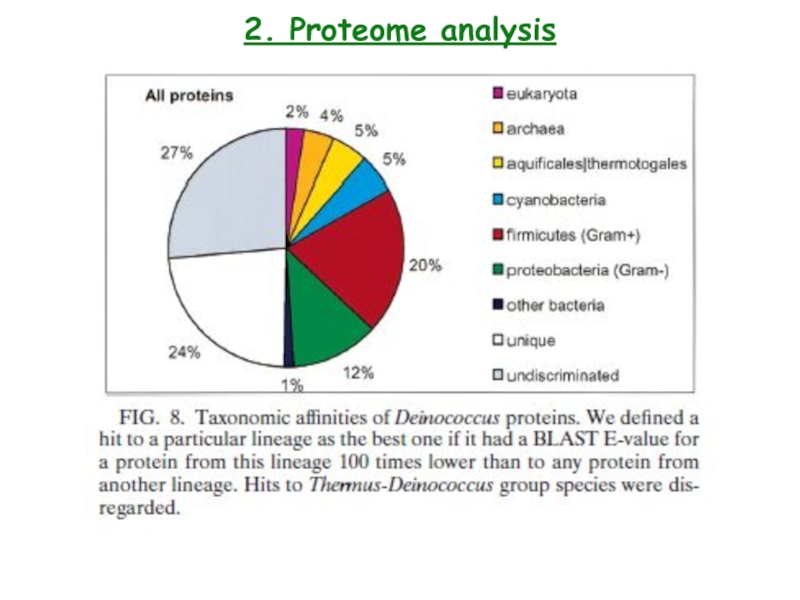
- 28. 2. Proteome analysis
- 29. 2. Proteome analysis Deinococcus radiodurans contains two thioredoxins
- 30. 2. Proteome analysis3. E.coli was cultures on
- 31. Reference Hitoshi Ito, Hiroshi Watanabe, Masaaki. Isolation
- 32. Reference John R. Battista, Mie-Jung Park, and
- 33. Thanks for attention
- 34. Скачать презентанцию
Слайды и текст этой презентации
Слайд 2Content
PHYSIOLOGICAL STUDIES
1. Nutritional studies
2. Growth studies
II.
GENETIC SYSTEMS
analysisСлайд 3Introduction
Deinococcus radiodurans is an extremophilic bacterium, one of the most radiation-resistant organisms known. It can
survive cold, dehydration, vacuum, acid and has been listed as the
world's toughest bacterium in The Guinness Book Of World Records. Also, they are mesophiles.*Gram-positive bacteria (* although its cell envelope is unusual and is reminiscent of the cell walls of Gram negative bacteria)
*Colonies- convex, smooth,
pink to red in color
(deinocrates - carotene)
* Habitat - rich in organic materials, such as soil, feces,
*Size of cells- 1.5 to 3.5 µm.
*Do not form endospores, non-motile
*Obligate aerobic chemo-organo-heterotroph
meat, or sewage, but has also been isolated from dried foods, room dust, medical instruments and textiles
Слайд 41. Nutritional studies: biochemical characteristics
(+++)-strong, (++)-moderate, (+)- weak, (-)-negative
Слайд 111. Nutritional studies: nutrition medium
Media requirements - simple in composition,
increase the yield of biomass.
Слайд 121. Nutritional studies: nutrition medium selection
3 strains of microorganisms: Deinococcus
radiodurans VKPM B-8209, Deinococcus radiodurans BKM-1422, Deinococcus radiodurans BKM-1467.
Different concentrations
of soy flour – 10g, 50g, 100g per liter
Strains of Deinococcus radiodurans was grown in 250-ml wide-mouthed Erlenmeyer flasks with 50 ml of this liquid media, which contributed 50 µl of overnight culture of bacteria.
Regime: 150 rpm on the circular shaker at 30°C for 48 hours
Evaluation of growth:
1) Visual: pink-orange color of medium (intensity of color)
2) Quantitative: number of CFU/ml (standard technique of parallel dilutions in saline solution and in-depth inoculation on solid agar medium [30°C for 24-48 hours])
Слайд 15Cultivation -> Solid state/submerged
Bioreactor design ->Airlift bioreactor
Fermentation mode ->
Batch, Continuous, Semi-continuous
Culture monitoring -> Primarily Visual evaluation (pink-orange
to red color and turbidity), microscopy, evaluation of cell density.Basic parameters->
*aerobic cultivation
* pH neutral 6,8-7,2 +/- 0.2 range
* Temperature 25°C- 37°C
*Stirring intensity and etc
2. Growth studies
Слайд 162. Genomic approaches
The genome of D. radiodurans consists of four major parts.
The complete sequence of the R1 strain has 3,284,156 base
pairs made up of two circular chromosomes (2,648,638 and 412,348 base pairs), a major plasmid (177,466 base pairs), and a small plasmid (45,704 base pairs).No current research shows whether or not these plasmids contribute specifically to functionality or virulence. However, it is known that multiple copies of each gene are found on all the chromosomes and plasmids, which most likely contributes to its amazing repair capabilities associated with its radiation resistance.
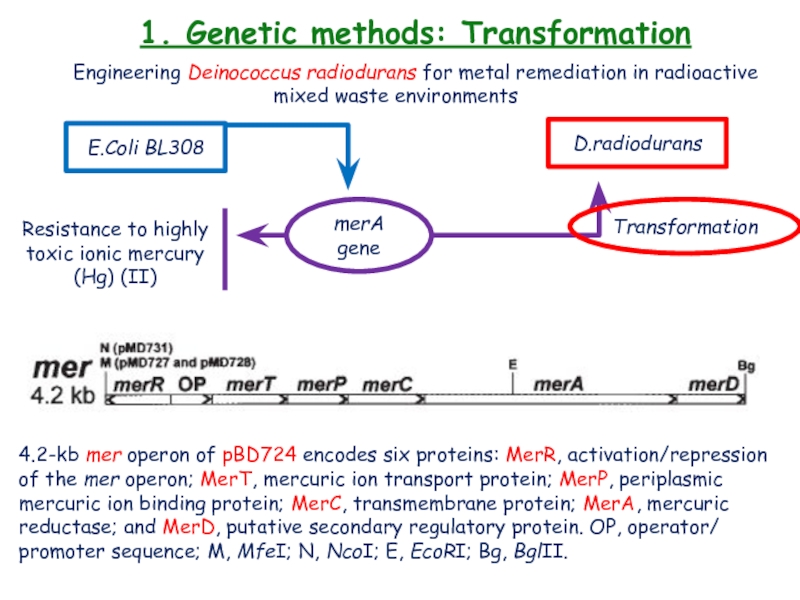
Слайд 191. Genetic methods: Transformation
Engineering Deinococcus radiodurans for metal remediation in
radioactive mixed waste environments
E.Coli BL308
D.radiodurans
merA gene
Resistance to
highly toxic ionic mercury (Hg) (II)Transformation
4.2-kb mer operon of pBD724 encodes six proteins: MerR, activation/repression of the mer operon; MerT, mercuric ion transport protein; MerP, periplasmic mercuric ion binding protein; MerC, transmembrane protein; MerA, mercuric reductase; and MerD, putative secondary regulatory protein. OP, operator/ promoter sequence; M, MfeI; N, NcoI; E, EcoRI; Bg, BglII.
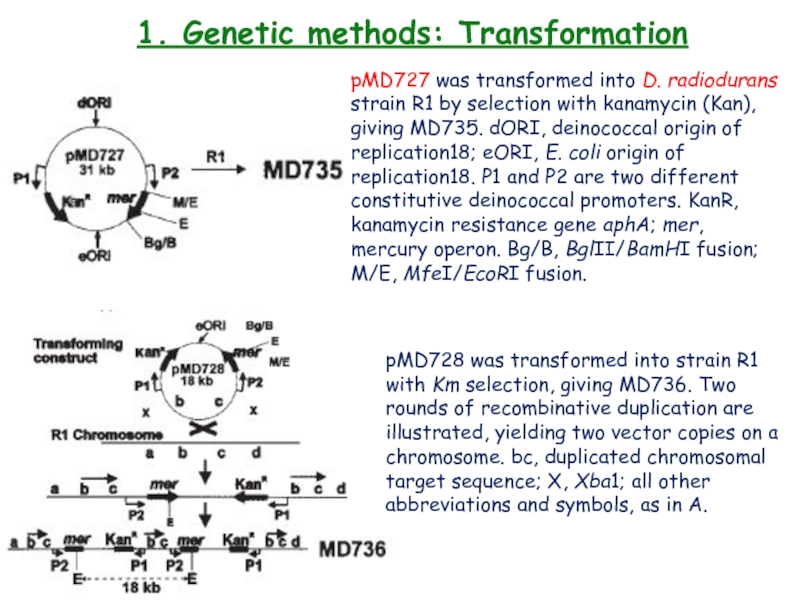
Слайд 201. Genetic methods: Transformation
pMD727 was transformed into D. radiodurans
strain R1 by selection with kanamycin (Kan), giving MD735. dORI,
deinococcal origin ofreplication18; eORI, E. coli origin of replication18. P1 and P2 are two different constitutive deinococcal promoters. KanR, kanamycin resistance gene aphA; mer, mercury operon. Bg/B, BglII/BamHI fusion; M/E, MfeI/EcoRI fusion.
pMD728 was transformed into strain R1
with Km selection, giving MD736. Two rounds of recombinative duplication are illustrated, yielding two vector copies on a chromosome. bc, duplicated chromosomal target sequence; X, Xba1; all other abbreviations and symbols, as in A.
Слайд 211. Genetic methods: Transformation
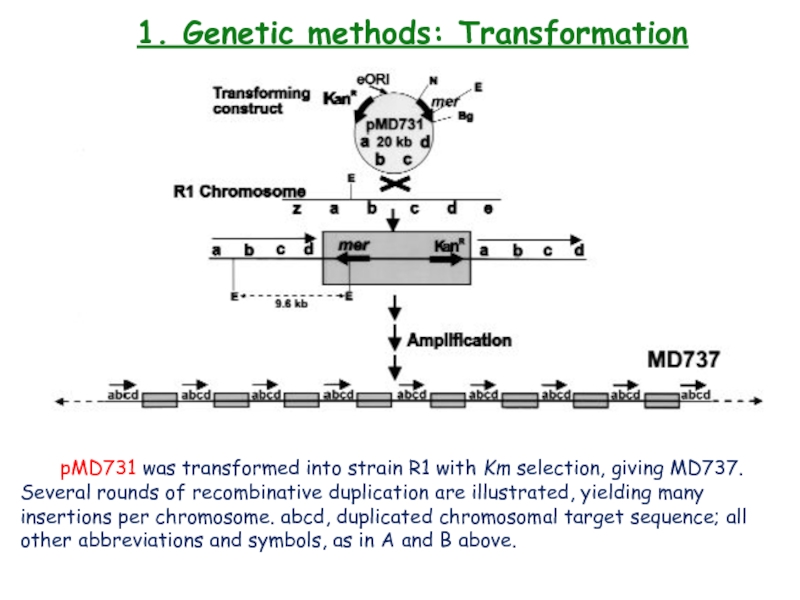
pMD731 was transformed into strain R1
with Km selection, giving MD737. Several rounds of recombinative duplication
are illustrated, yielding many insertions per chromosome. abcd, duplicated chromosomal target sequence; all other abbreviations and symbols, as in A and B above.Слайд 221. Genetic methods: Transformation
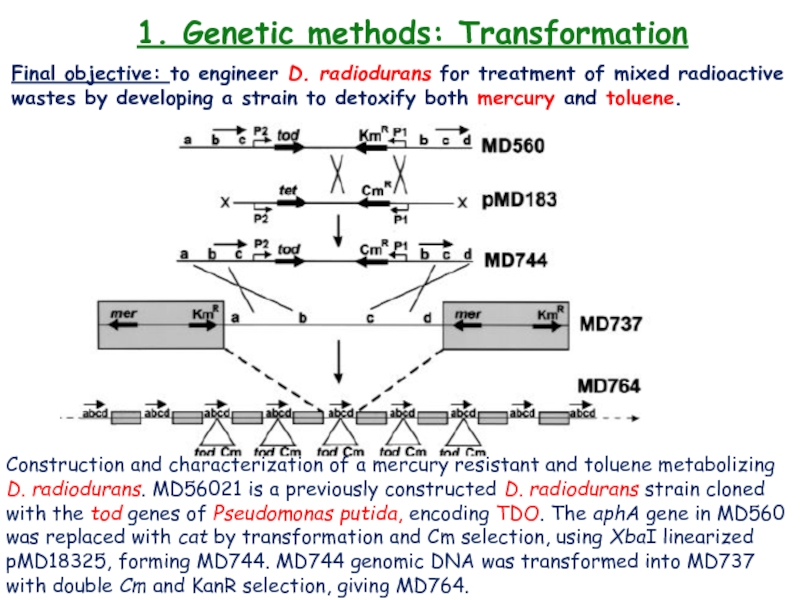
Final objective: to engineer D. radiodurans
for treatment of mixed radioactive wastes by developing a strain
to detoxify both mercury and toluene.Construction and characterization of a mercury resistant and toluene metabolizing D. radiodurans. MD56021 is a previously constructed D. radiodurans strain cloned with the tod genes of Pseudomonas putida, encoding TDO. The aphA gene in MD560 was replaced with cat by transformation and Cm selection, using XbaI linearized pMD18325, forming MD744. MD744 genomic DNA was transformed into MD737 with double Cm and KanR selection, giving MD764.
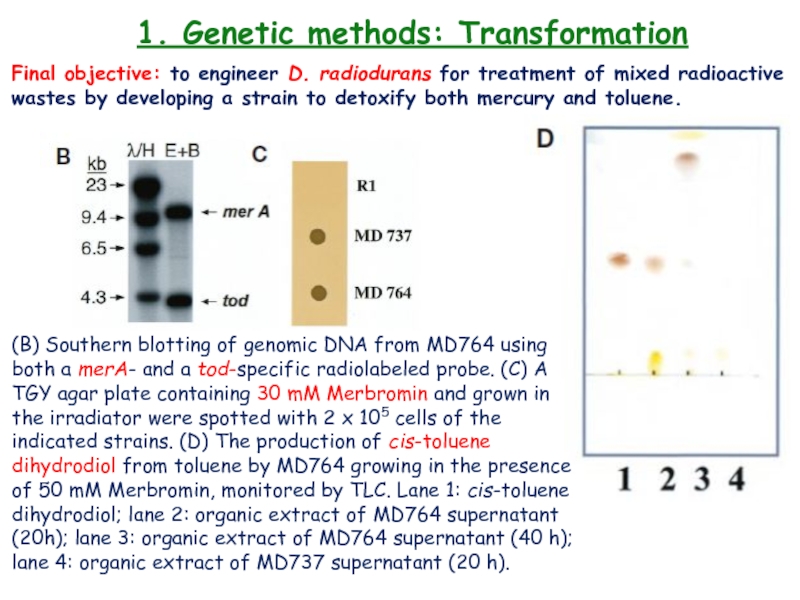
Слайд 231. Genetic methods: Transformation
Final objective: to engineer D. radiodurans
for treatment of mixed radioactive wastes by developing a strain
to detoxify both mercury and toluene.(B) Southern blotting of genomic DNA from MD764 using both a merA- and a tod-specific radiolabeled probe. (C) A TGY agar plate containing 30 mM Merbromin and grown in the irradiator were spotted with 2 x 105 cells of the indicated strains. (D) The production of cis-toluene dihydrodiol from toluene by MD764 growing in the presence of 50 mM Merbromin, monitored by TLC. Lane 1: cis-toluene dihydrodiol; lane 2: organic extract of MD764 supernatant (20h); lane 3: organic extract of MD764 supernatant (40 h); lane 4: organic extract of MD737 supernatant (20 h).
Слайд 241. Genetic methods: mobile elements
Inteins. Two inteins, protein splicing elements
that are typically inserted in genes involved in DNA metabolism
and other nucleotide-utilizing enzymes.One of these is inserted in the ribonucleotide reductase and is similar to the inteins inserted in orthologous enzymes from B. subtilis, pyrococci, and chilo iridiscent virus.
The second intein is inserted between the P-loop motif and the Mg2+-binding (Walker B) motif of a SWI2/SNF2 family ATPase, which is involved in chromatin remodeling; this is the first documented instance of an intein interrupting a protein of this family.
Слайд 251. Genetic methods: mobile elements
Overall, 52 IS elements were detected
in the D. radiodurans genome (Table 7). The three most
abundant ISs are IS4_DR (13 copies), IS2621_DR (11 copies), and IS200_DR (8 copies). IS elements are unevenly distributed on the chromosomes and plasmids. The number of copies per 10,000 nucleotides in the plasmid and the megaplasmid is more than 10 times greater than the number found in chromosomes I and II.There is, however, little direct evidence for any active transposition in D.radiodurans. In the entire genome, there is only one example of gene disruption by an IS element, where IS2621 is inserted into the gene for alkaline serine exoprotease A (aqualysin I).
Слайд 261. Genetic methods: mobile elements
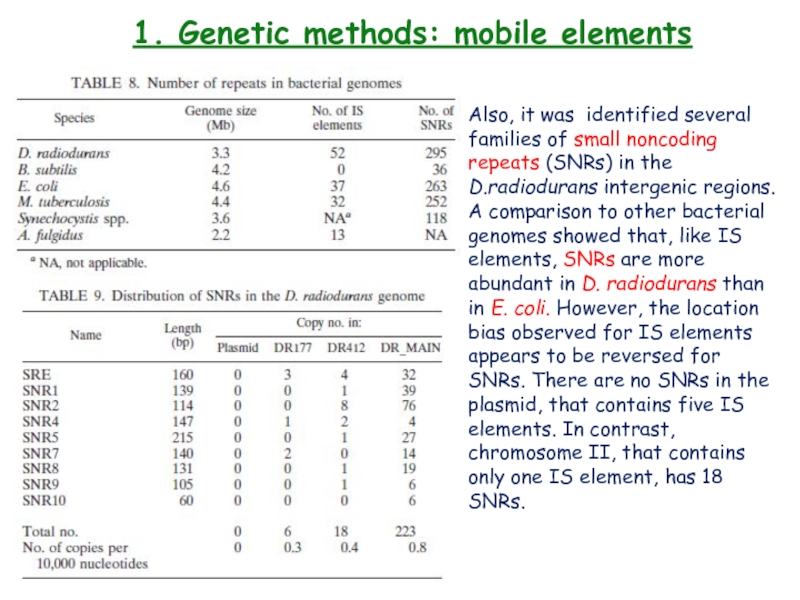
Also, it was identified several families
of small noncoding repeats (SNRs) in the D.radiodurans intergenic regions.
A comparison to other bacterial genomes showed that, like IS elements, SNRs are more abundant in D. radiodurans than in E. coli. However, the location bias observed for IS elements appears to be reversed for SNRs. There are no SNRs in the plasmid, that contains five IS elements. In contrast, chromosome II, that contains only one IS element, has 18 SNRs.Слайд 271. Genetic methods: Gene inactivation
Inactivation of Proteins Presumed to Be
Involved in the Desiccation Tolerance
Mutational inactivation of the genes designated
DR1172 and DRB0118 in Deinococcus radiodurans R1 greatly sensitizes this species to desiccation, but not to ionizing radiation. These genes encode proteins that share features with the desiccation-induced LEA76 proteins of many plants and the PCC13-62 protein of Craterostigma plantagineum, suggesting that D. radiodurans may serve as a useful model for the study of desiccation tolerance in higher organisms.Inactivation method- In vitro transposition was performed using the protocol developed by New England Biolabs Beverly,
-> circular pGTC101 was combined with the TnsABC* transposase supplied with the system and target DNA. The vector pGTC101 carries the transposon TnDrCat. When combined with this transposase, the transposon excises from pGTC101 and inserts randomly into the target DNA. The transposition reaction mixture was transformed into targeted cells by electroporation.
-> then colonies with insertion are selected, and the desiccation impact is tested.
Слайд 292. Proteome analysis
Deinococcus radiodurans contains two thioredoxins (Trx and Trx1)
and a single thioredoxin reductase (TrxR) as part of its
response to oxidative stress. Thioredoxin reductase is a member of the family of pyridine nucleotide-disulfide oxidoreductase flavoenzymes.Steps:
1. The TrxR gene (DR1982) was obtained by the polymerase chain reaction (PCR) using D. radiodurans strain R1 genomic DNA as a template (ATCC). The following were used as forward and reverse oligonucleotide primers for PCR, respectively: 5’-GCG CCA TGG GTA TGA CGG CAC CTA CTG-3’ and 5’-GCG GGA TCC TCA GTC GGC AGCC-3’.
2. 1kbp fragment was purified by gel extraction, digested with NcoI and BamHI and cloned into a modified pET-30b vector, by which E.coli is transformed for cloning.
Слайд 302. Proteome analysis
3. E.coli was cultures on LB agar plates
with kanamycin, Then single colonies were selected and grown overnight
in LB media with also kanamycin.4. The frozen cell pellets were thawed and resuspended in sonication buffer (50 mM Tris–HCl pH 8.0, 1 mM AEBSF, 20 mg ml1 Dnase and 20 mg ml1 lysozyme).
The thawed cells were mechanically disrupted by sonication and cell debris was removed by centrifugation. The resulting supernatant was loaded onto a POROS MC50 metal-chelation column (Applied Biosystems,USA) pre-equilibrated with a buffer containing 5 mM imidazole, 0.5 M NaCl, 50 mM Tris– HCl pH 8.0. The column was washed with a 60 mM imidazole, 0.5 M NaCl, 50 mM Tris–HCl pH 8.0 buffer to remove non-specific binding and the protein eluted with a 0–1 M imidazole gradient. Fractions (10 ml) were collected and the purity of the protein was checked on Coomassie-stained SDS–PAGE.
Слайд 31Reference
Hitoshi Ito, Hiroshi Watanabe, Masaaki. Isolation and Identification of
Radiation-resistant Cocci Belonging to the Genus Deinococcus, Agric. Biol Chern.,
47 (6), 1239-1247, 1983KIRA S. MAKAROVA et al. Genome of the Extremely Radiation-Resistant Bacterium Deinococcus radiodurans Viewed from the Perspective of Comparative Genomics. MICROBIOLOGY AND MOLECULAR BIOLOGY REVIEWS, Mar. 2001, Vol. 65, No. 1, p. 44–79.
Ronald M. Atlas. Handbook of microbiological media. 4th editijn. ASM press Washington D.C. © 2010 by Taylor and Francis Group, LLC
White, O. et al. Genome sequence of the radioresistant bacterium Deinococcus radiodurans R1. Science 286, 1571-1577 (November 19, 1999).
Hassan Brim, Sara C. McFarlan, James K. Fredrickson, Kenneth W. Minton, Min Zhai, Lawrence P. Wackett, and Michael J. Daly. Engineering Deinococcus radiodurans for metal remediation in radioactive mixed waste environments. NATURE BIOTECHNOLOGY VOL 18 JANUARY 2000
Слайд 32Reference
John R. Battista, Mie-Jung Park, and Andrew E. McLemore.
Inactivation of Two Homologues of Proteins Presumed to Be Involved
in the Desiccation Tolerance of Plants Sensitizes Deinococcus radiodurans R1 to Desiccation. Cryobiology 43, 133–139 (2001)Josiah Obiero, Sara A. Bonderoff, Meghan M. Goertzen and David A. R. Sanders. Expression, purification, crystallization and preliminary X-ray crystallographic studies of Deinococcus radiodurans thioredoxin reductase. Acta Cryst. (2006). F62, 757–760
Links- https://en.wikipedia.org/wiki/Deinococcus_radiodurans
http://russianpatents.com/patent/241/2418061.html