Разделы презентаций
- Разное
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Математика
- Медицина
- Менеджмент
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, картинки для презентаций
- Экология
- Экономика
- Юриспруденция
THERMAL PHYSICS
Содержание
- 1. THERMAL PHYSICS
- 2. Learning Objectivesunderstand and apply first law of
- 3. Terms to Remember!ThermodynamicsThermodynamic SystemSurroundingsHeatWorkInternal Energy
- 4. ThermodynamicsThermodynamics is the macroscopic study of the
- 5. Thermodynamic SystemThermodynamic System is a macroscopic aspect
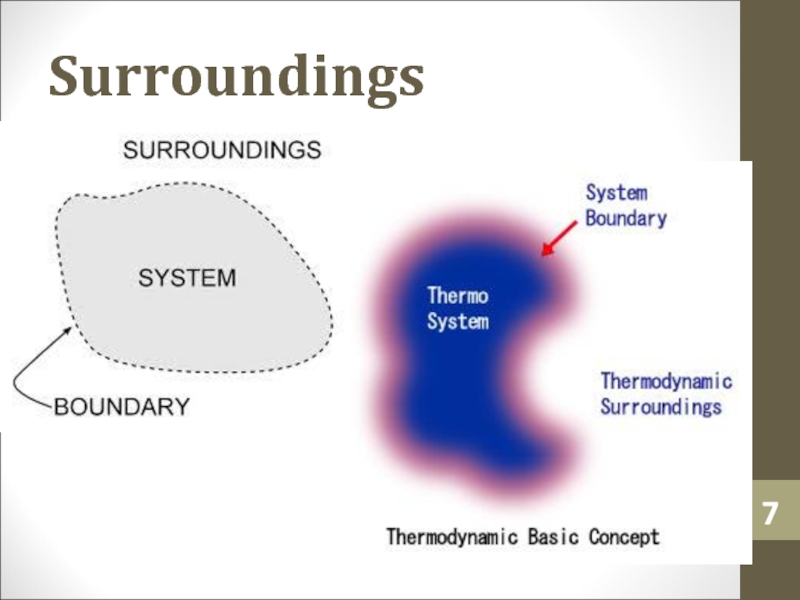
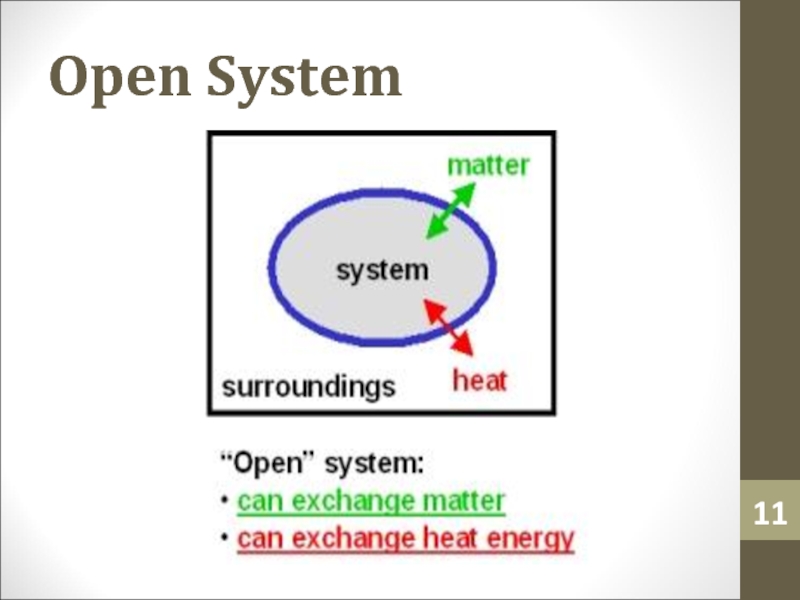
- 6. SurroundingsSurroundings is everything in the problem outside
- 7. Surroundings
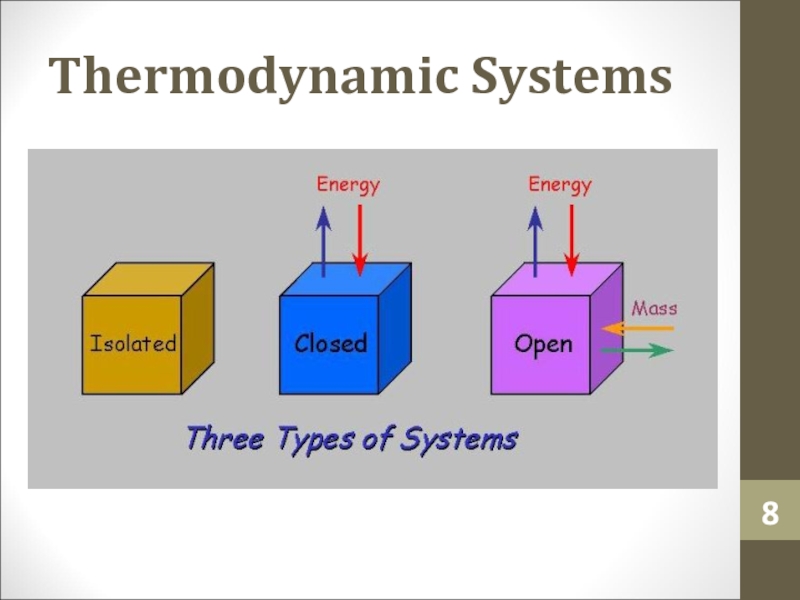
- 8. Thermodynamic Systems
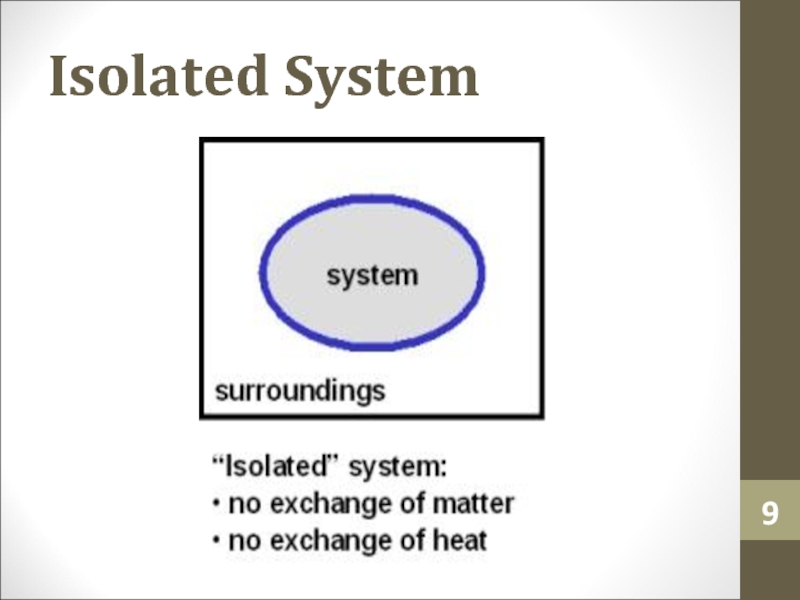
- 9. Isolated System
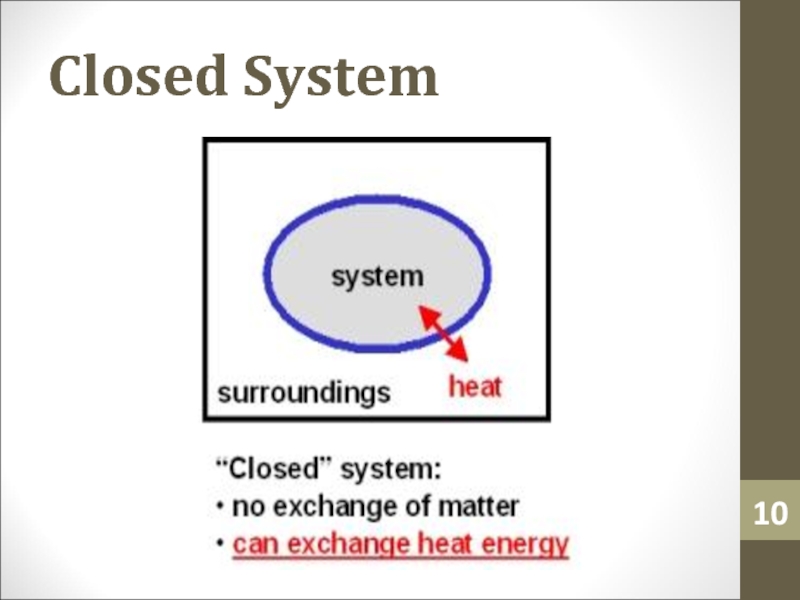
- 10. Closed System
- 11. Open System
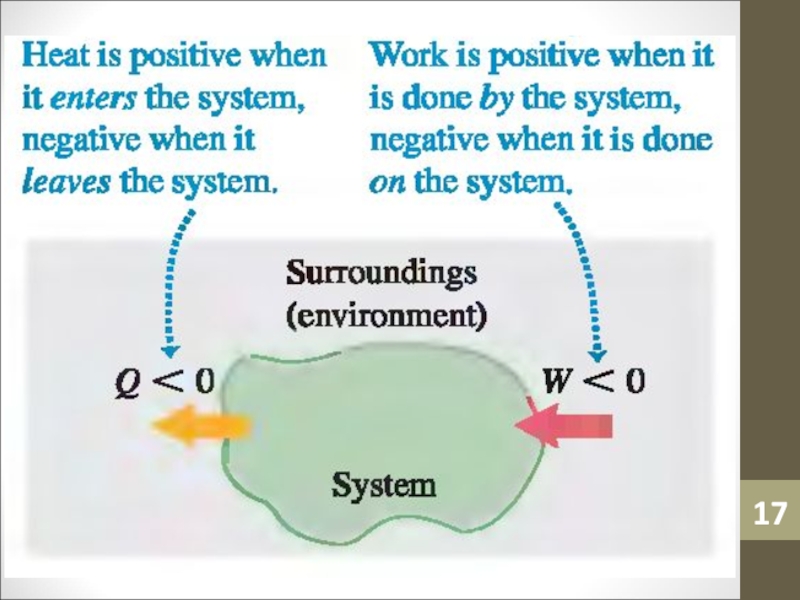
- 12. HeatHeat (Q) is an amount of thermal
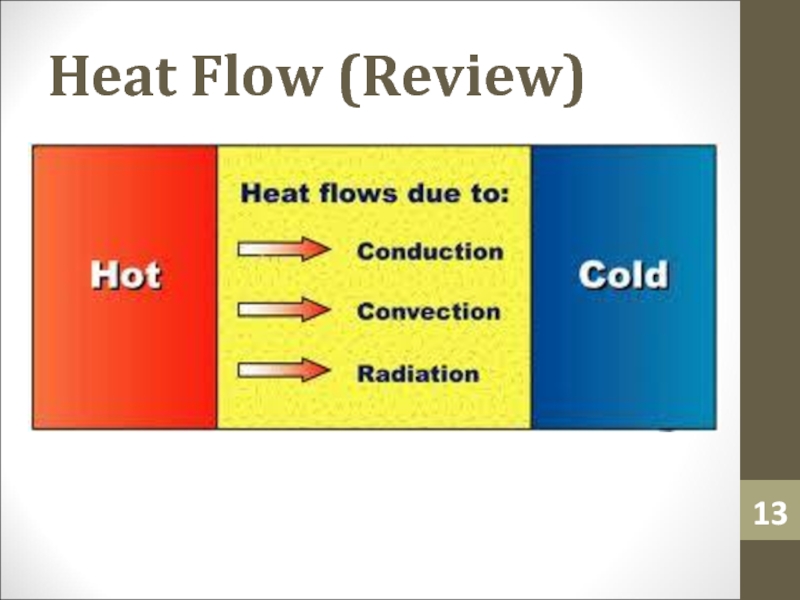
- 13. Heat Flow (Review)
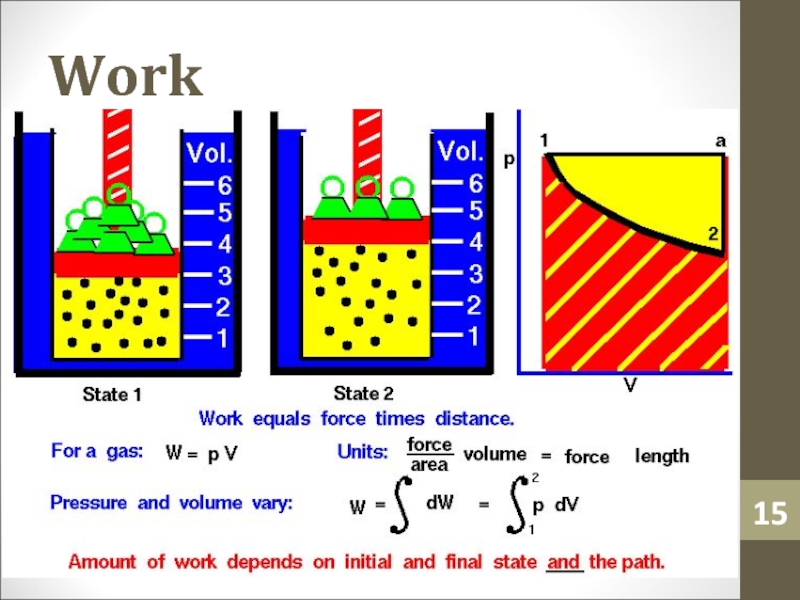
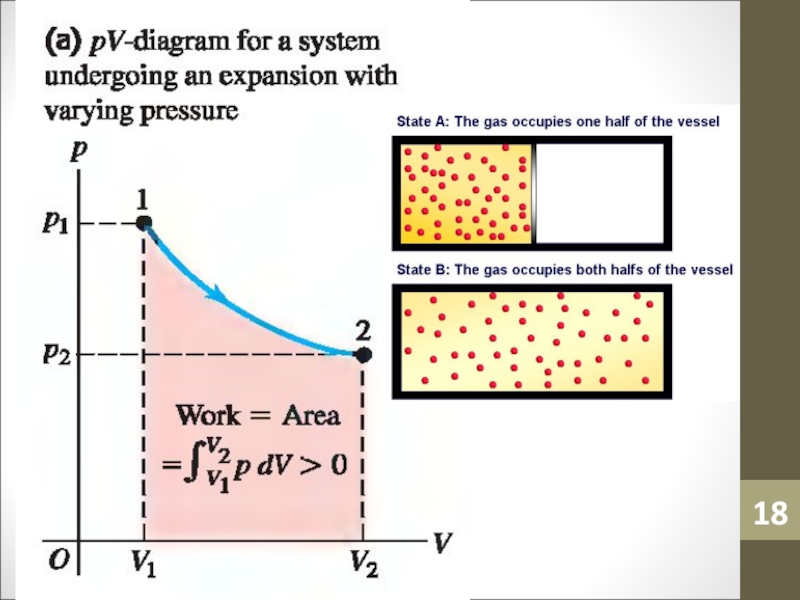
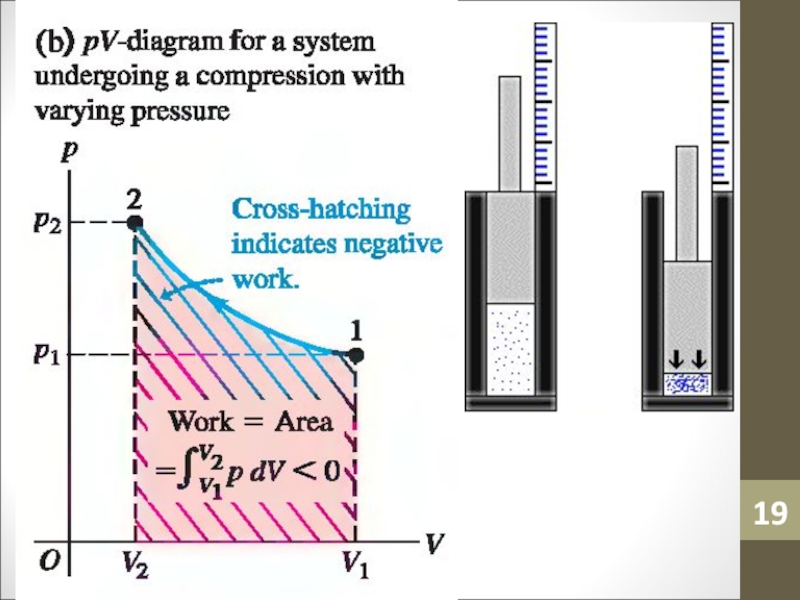
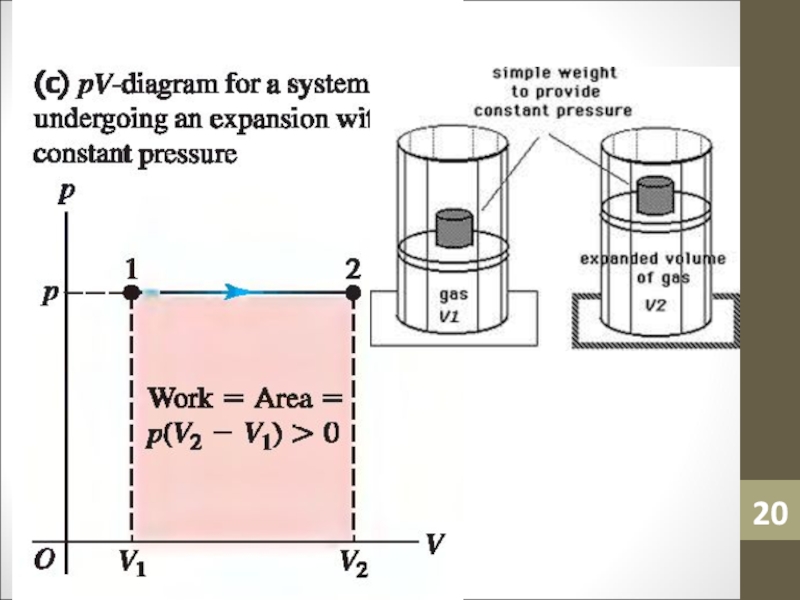
- 14. WorkWork (W) is simply a macroscopic transfer of energy from the gas to the surroundings.
- 15. Work
- 16. Слайд 16
- 17. Слайд 17
- 18. Слайд 18
- 19. Слайд 19
- 20. Слайд 20
- 21. Change in Internal EnergyChange in Internal Energy
- 22. Change in Internal EnergyThe change in internal
- 23. 1st Law of ThermodynamicsThe First Law of
- 24. 1st Law of ThermodynamicsSince Q and W
- 25. 1st Law of ThermodynamicsThe internal energy of
- 26. StateState is defined as the physical condition of the system.
- 27. Important NoteA given system at any moment
- 28. Important NoteRather, when work is done on
- 29. Important NoteThus, work and heat are involved
- 30. State VariablesQuantities which describe the state of
- 31. Sample Problem2500 J of heat is added
- 32. Sample Problem2500 J of heat is added
- 33. Скачать презентанцию
Learning Objectivesunderstand and apply first law of thermodynamicsdistinguish graphs of adiabatic and isothermal processesunderstand second law of thermodynamics
Слайды и текст этой презентации
Слайд 2Learning Objectives
understand and apply first law of thermodynamics
distinguish graphs of
adiabatic and isothermal processes
Слайд 4Thermodynamics
Thermodynamics is the macroscopic study of the behaviour of systems.
It was a mathematical theory developed before a detailed understanding
of the particulate nature of gases.Слайд 5Thermodynamic System
Thermodynamic System is a macroscopic aspect of a problem
than can be considered as a separate whole. An ideal
gas for example can have energy flowing in or out of it fromthe surroundings.
Слайд 6Surroundings
Surroundings is everything in the problem outside the
thermodynamic system.
Heat can flow from the system to the surroundings and
vice versa.Слайд 12Heat
Heat (Q) is an amount of thermal energy transferred from
the surroundings to an ideal gas. It is a result
of a temperature difference.Слайд 21Change in Internal Energy
Change in Internal Energy (∆U) is the
change in a gas’ energy due to the PE and
KE of the molecules.It does not include external factors such as gravity.
∆U for an ideal gas will involve temperature changes (∆θ).
Слайд 22Change in Internal Energy
The change in internal energy (ΔU) of
a closed system will be equal to the energy added
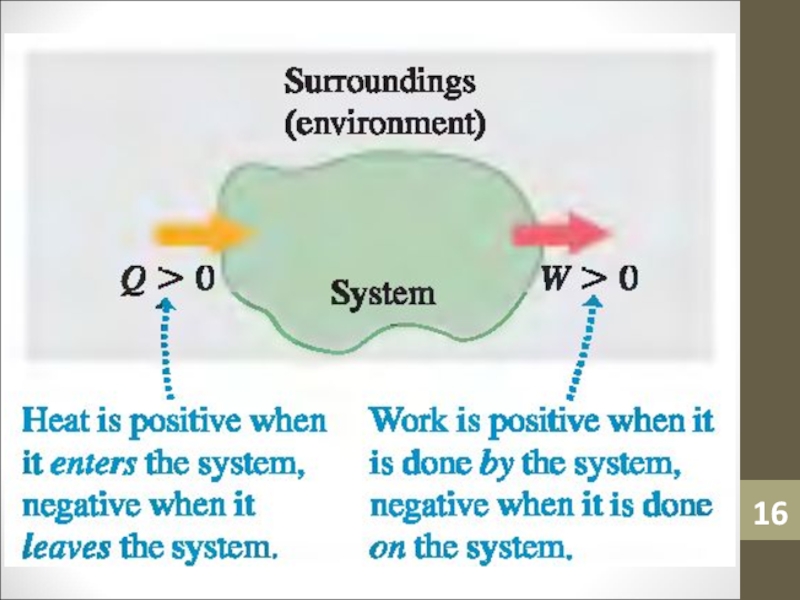
to the system by heating minus the work done by the system on the surroundings.ΔU = Q – W
Слайд 231st Law of Thermodynamics
The First Law of Thermodynamics states that
when heat Q is added to a system while the
system does work W, the internal energy U changes by an amount equal to Q – W.ΔU = Q – W
Слайд 241st Law of Thermodynamics
Since Q and W represent energy transferred
into or out of the system , the internal energy
changes accordingly.The First Law of Thermodynamics is great and broad statement of the law of conservation of energy.
Слайд 251st Law of Thermodynamics
The internal energy of any thermodynamic system
depends only on its state. The change in internal energy
in any process depends only on the initial and final states, not on the path.The internal energy of an isolated system is constant.
Слайд 27Important Note
A given system at any moment is in particular
state and can be said to have a certain amount
of internal energy.But a system does NOT have a certain amount of heat or work.
Слайд 28Important Note
Rather, when work is done on a system or
when heat is added or removed from a system, the
state of the system changes.Слайд 29Important Note
Thus, work and heat are involved in thermodynamic processes
that can change the system from one state to another;
they are not characteristic of the state itself.Слайд 30State Variables
Quantities which describe the state of the system is
called state variables.
Internal Energy (U) Mass (m)
Pressure (P) Volume (V)
Temperature (T)
Number of Moles (n)