Разделы презентаций
- Разное
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Математика
- Медицина
- Менеджмент
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, картинки для презентаций
- Экология
- Экономика
- Юриспруденция
LECTURE № 11 ELECTROCHEMICAL PROCESSES 1 8.04.2017
Содержание
- 1. LECTURE № 11 ELECTROCHEMICAL PROCESSES 1 8.04.2017
- 2. Today’s objectives: Define electrode, anode, cathode, anion,
- 3. REMINDER:“Redox” Chemistry: Reduction and Oxidation reactions are
- 4. Electrochemistry is the branch of science which
- 5. Electrochemical Reaction:Redox (oxidation-reduction) reactions in which electrons
- 6. Relating electricity and chemical reactionsTransfer of electronsGalvanic
- 7. In pure solid metal ion - atom
- 8. Metal surfaceWater
- 9. (-) Ме ANODE(+) Ме CATHODEМе-----++++If metal is
- 10. An electrode in an electrochemical cell is referred to
- 11. The layer of positive / negative ions
- 12. Standard electrode potential (SEP) is a measure
- 13. Measurement of SEP SEP cannot be measured directly.
- 14. Type of electrode: Gas electrode (Primary Reference
- 15. STANDARD HYDROGEN ELECTRODE (SHE) is compared the
- 16. Cathode:Cu2+ +2e = CuAnode:H2 – 2e = 2H+Н2Voltmeter: E=+0.34V
- 17. Слайд 17
- 18. Saturated Calomel Electrode (SCE)Type/class: Metal-metal insoluble salt
- 19. Слайд 19
- 20. In electrochemistry, for calculating electrode potential of
- 21. At any specific temperature, the Nernst equation
- 22. Problem:Calculate the electrode potential of zinc if
- 23. We know that reduction (gaining electrons) can’t
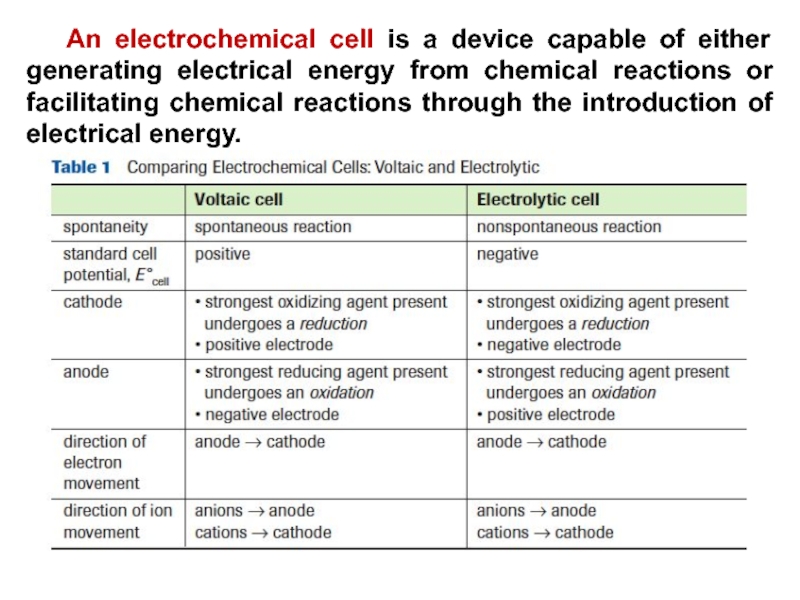
- 24. An electrochemical cell is a device capable
- 25. An electrichemical cell converts chemical energy into
- 26. Alessandro Volta’s invention was an immediate technological
- 27. A galvanic cell, or voltaic cell, named
- 28. Galvanic cell composed of two half-cells; which
- 29. The driving force which makes the electrons
- 30. If the E>0, it is positive, the reaction occurring is spontaneous. If the E
- 31. Слайд 31
- 32. Representation of a galvanic cellGalvanic cell consists
- 33. Many natural phenomena are based on electrochemical processes, such
- 34. Electric batteries use electrochemical processes to store
- 35. Скачать презентанцию
Слайды и текст этой презентации
Слайд 2Today’s objectives:
Define electrode, anode, cathode, anion, cation, salt bridge/porous
cup, electrolyte, and voltaic cell
that occurs at each electrode in an electrochemical cellСлайд 3REMINDER:
“Redox” Chemistry: Reduction and Oxidation reactions are all reactions that
involve the change of an oxidation number, and transfer of
electrons among the reacting substances.Oxidation: Loss of electrons (increase in oxidation number): Zn – 2e Zn2+
Reduction: Gain of electrons (a reduction in oxidation number): Cu2+ + 2e Cu
Electrons are transferred from the reducing agent (the species being oxidized) to the oxidizing agent (the species being reduced).
Слайд 4 Electrochemistry is the branch of science which deals with the
relationship between chemical reaction and electricity.
An electrochemical process is a chemical
reaction that either causes or is caused by the movement of electrical current. These processes are a type of oxidation-reduction reaction in which one atom or molecule loses an electron to another atom or molecule. In electrochemical reactions, the atoms or molecules in the reaction are relatively far apart from each other compared to other reactions, forcing the electrons being transferred to travel a greater distance and thereby produce an electrical current.
Слайд 5Electrochemical Reaction:
Redox (oxidation-reduction) reactions in which electrons are transferred from
a donor (reducing agent) to an acceptor (oxidant).
Redox reactions takes
place by movement of electrons or ions across the interface of metal electrode.Each of the reaction is known as half-reaction and system of an electrode with electrolyte is called half-cell.
A half-cell is a structure that contains a conductive electrode and a surrounding conductive electrolyte separated by a naturally occurring Helmholtz electrical double layer.
Both half-reactions must always go side by side to sustain the electrochemical reaction.
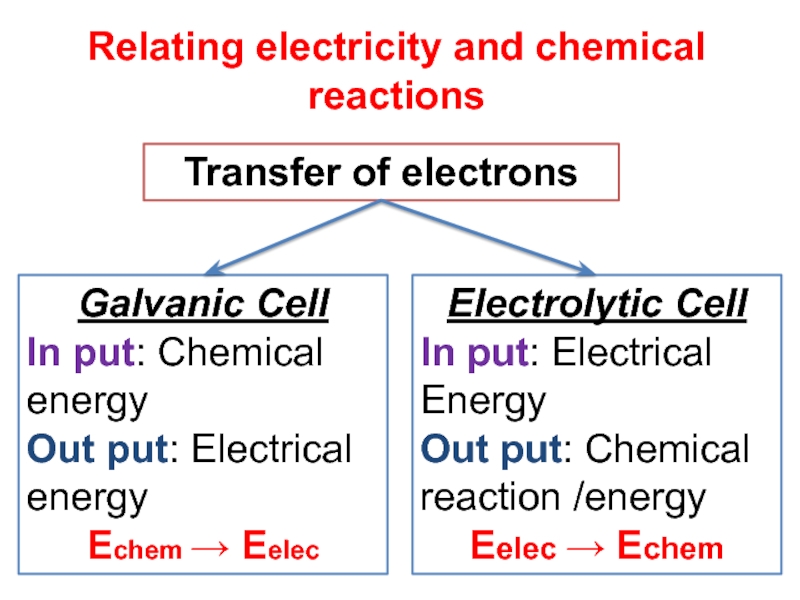
Слайд 6Relating electricity and chemical reactions
Transfer of electrons
Galvanic Cell
In put: Chemical
energy
Out put: Electrical energy
Echem Eelec
Electrolytic Cell
In put: Electrical Energy
Out
put: Chemical reaction /energyEelec Echem
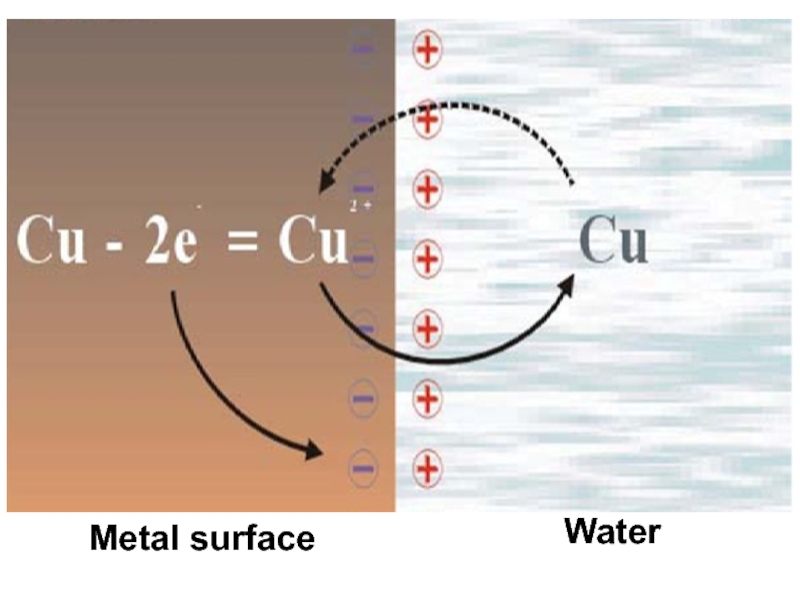
Слайд 7 In pure solid metal ion - atom located in the
sites of the crystal lattice, and are in equilibrium with
free electrons:Ме+n • ē Ме+n + nē
When a metal is immersed in the water in the system is established redox equilibrium:
Ме + mH2O Me (H2O)m + nē
n+
electrode solution electrode
Ме
WHAT IS A ELECTRODE?
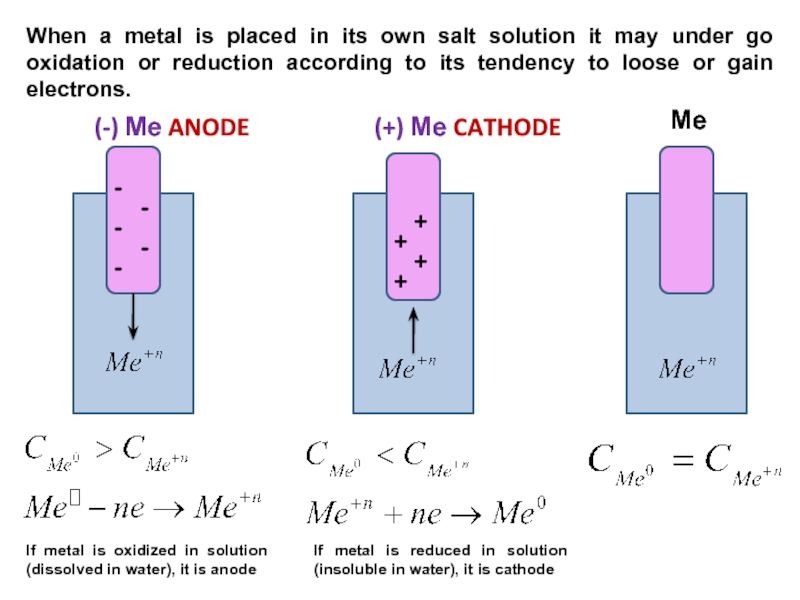
Слайд 9(-) Ме ANODE
(+) Ме CATHODE
Ме
-
-
-
-
-
+
+
+
+
If metal is oxidized in solution
(dissolved in water), it is anode
When a metal is placed
in its own salt solution it may under go oxidation or reduction according to its tendency to loose or gain electrons.If metal is reduced in solution (insoluble in water), it is cathode
Слайд 10 An electrode in an electrochemical cell is referred to as either an anode or
a cathode (words that were coined by William Whewell at Faraday's request).
Electrodes: are
usually metal strips/wires connected by an electrically conducting wire.Anode: is the electrode where oxidation takes place, it is the negative (-) electrode (example, active metals are soluble in water).
Cathode: is the electrode where reduction takes place, it is the positive (+) electrode (example, passive metals are insoluble in water).
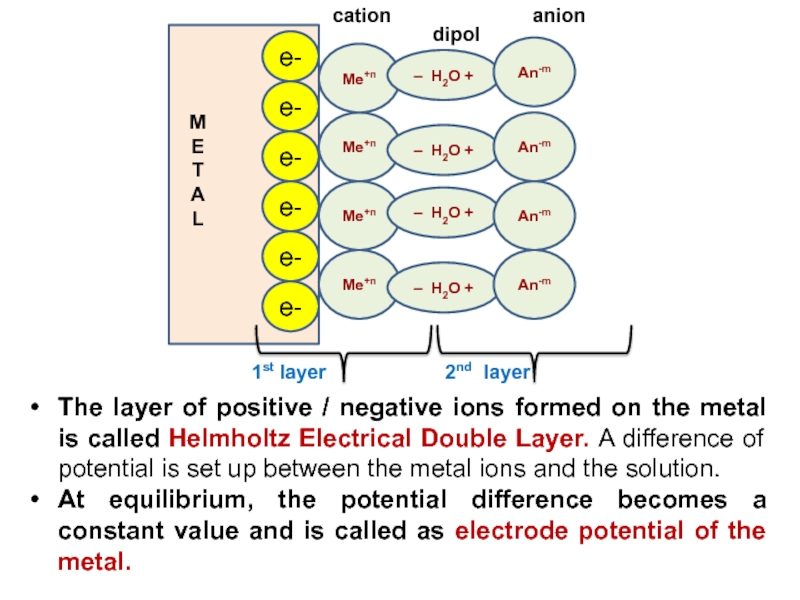
Слайд 11The layer of positive / negative ions formed on the
metal is called Helmholtz Electrical Double Layer. A difference of
potential is set up between the metal ions and the solution.At equilibrium, the potential difference becomes a constant value and is called as electrode potential of the metal.
e-
e-
e-
e-
e-
e-
Ме+n
Ме+n
Ме+n
Ме+n
– H2O +
– H2O +
– H2O +
– H2O +
An-m
An-m
An-m
An-m
1st layer
2nd layer
M
E
T
A
L
dipol
anion
cation
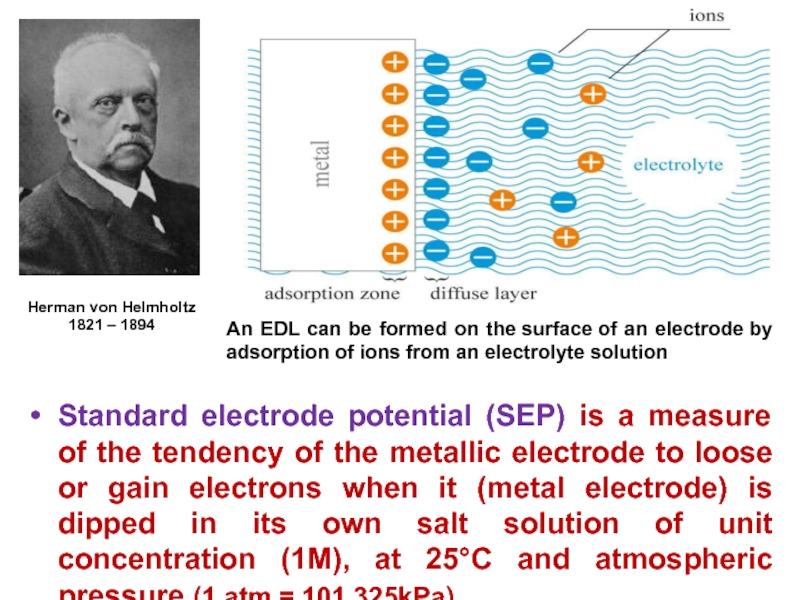
Слайд 12Standard electrode potential (SEP) is a measure of the tendency
of the metallic electrode to loose or gain electrons when
it (metal electrode) is dipped in its own salt solution of unit concentration (1M), at 25C and atmospheric pressure (1 atm = 101,325kPa).Herman von Helmholtz
1821 – 1894
An EDL can be formed on the surface of an electrode by adsorption of ions from an electrolyte solution
Слайд 13Measurement of SEP
SEP cannot be measured directly. The electrode is
coupled with a reference electrodes:
Standard Hydrogen electrode (SHE)
Saturated Calomel Electrode
(SCE)Reference electrode is an electrode which has a stable electrode potential and with which we can compare the potentials of other electrodes.
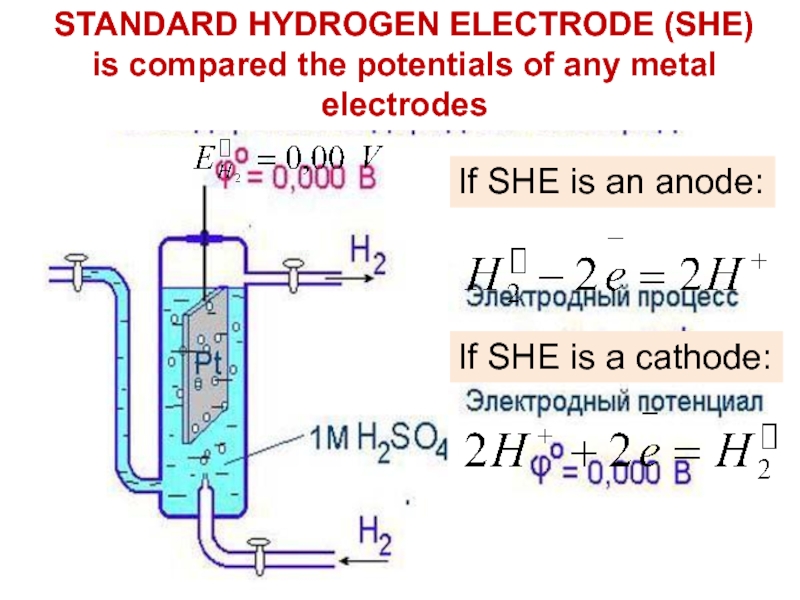
Слайд 14Type of electrode: Gas electrode (Primary Reference Electrode)
Components:
Electrode component:
Pt – H2
Electrolyte component: H2SO4 (1M)
Electrode representation:
Pt, H2 (1atm) /
2H+ (1M)Construction: Hydrogen electrode consists of a Platinum foil connected to a platinum wire sealed in a glass tube. The electrode is in contact with 1M H2SO4 and hydrogen gas (1 atmosphere) is constantly bubbled.
Limitations
• It requires pure hydrogen gas and is difficult to set up and to transport
• It requires large volume of test solution
• The potential of the electrode is dependent on atmospheric pressure
STANDARD HYDROGEN ELECTRODE (SHE)
Слайд 15STANDARD HYDROGEN ELECTRODE (SHE) is compared the potentials of any
metal electrodes
If SHE is an anode:
If SHE is a cathode:
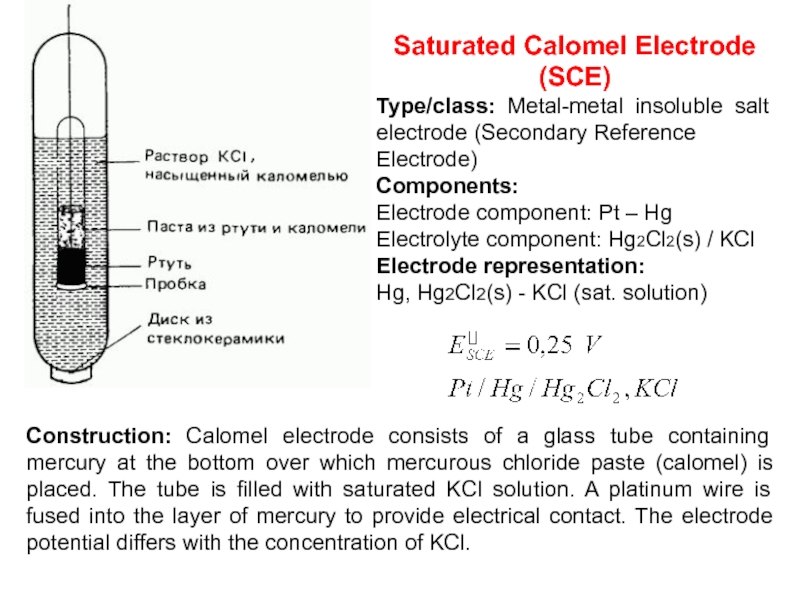
Слайд 18Saturated Calomel Electrode (SCE)
Type/class: Metal-metal insoluble salt electrode (Secondary Reference
Electrode)
Components:
Electrode
component: Pt – Hg
Electrolyte component: Hg2Cl2(s) / KCl
Electrode representation:
Hg, Hg2Cl2(s)
- KCl (sat. solution)Construction: Calomel electrode consists of a glass tube containing mercury at the bottom over which mercurous chloride paste (calomel) is placed. The tube is filled with saturated KCl solution. A platinum wire is fused into the layer of mercury to provide electrical contact. The electrode potential differs with the concentration of KCl.
Слайд 20 In electrochemistry, for calculating electrode potential of half-cell used the
Nernst equation. This equation is relates the electrode potential of
a half-cell at any point in time to the standard electrode potential, temperature, activity, and reaction quotient of the underlying reactions and species:EMe is the half-cell metal electrode potential at the temperature of interest
EoMe is the standard half-cell electrode potential
R is the universal gas constant: R = 8.314 472(15) J *K−1 *mol−1
T is the absolute temperature
a is the chemical activity for the relevant species, activities in the Nernst equation are frequently replaced by simple concentrations.)
F is the Faraday constant: F = 9.648 533 99(24)×104 C mol−1
z is the number of moles of electrons transferred in the cell reaction or half-reaction
Слайд 21 At any specific temperature, the Nernst equation derived above can
be reduced into a simple form. For example, at room
temperature (25 °C), RT/F may be treated like a constant and replaced by 25.693 mV for cells. The Nernst equation is frequently expressed in terms of base 10 logarithms (i.e., common logarithms) rather than natural logarithms, in which case it is written, for a cell at 25 °C:Слайд 22Problem:
Calculate the electrode potential of zinc if it was dipped
in 0,01M ZnSO4 solution
Using the Nernst equation:
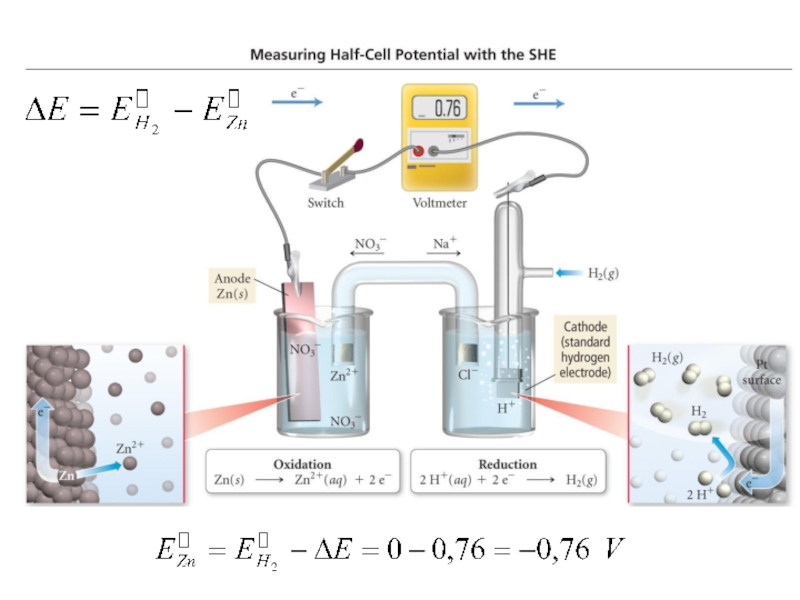
Слайд 23 We know that reduction (gaining electrons) can’t happen without an
oxidation to provide the electrons.
When two half-cells (metal electrodes) are
joined by a salt bridge or some other path (porous membrane) that allows ions to pass between the two sides in order to maintain electro neutrality are obtained electrochemical cell, where oxidation occurs at one half cell while reduction takes place at the other half cell.Слайд 24 An electrochemical cell is a device capable of either generating
electrical energy from chemical reactions or facilitating chemical reactions through
the introduction of electrical energy.Слайд 25An electrichemical cell converts chemical energy into electrical energy
Alessandro Volta
invented the first electric cell but got his inspiration from
Luigi Galvani. Galvani’s crucial observation was that two different metals could make the muscles of a frog’s legs twitch. Unfortunately, Galvani thought this was due to some mysterious “animal electricity”. It was Volta who recognized this experiment’s potential.An electric cell produces very little electricity, so Volta came up with a better design:
A battery is defined as two or more electric cells connected in series to produce a steady flow of current
Volta’s first battery consisted of several bowls of brine (NaCl(aq)) connected by metals that dipped from one bowl to another
His revised design, consisted of a sandwich of two metals separated by paper soaked in salt water.
Слайд 26Alessandro Volta’s invention was an immediate technological success because it
produced electric current more simply and reliably than methods that
depended on static electricity.It also produced a steady electric current –something no other device could do.
Luigi Galvani
Alessandro Volta
Слайд 27 A galvanic cell, or voltaic cell, named after Luigi Galvani,
or Alessandro Volta respectively, is an electrochemical cell that derives
electrical energy from spontaneous redox reactions taking place within the cell. It generally consists of two different metals connected by a salt bridge, or individual half-cells separated by a porous membrane.Galvanic or voltaic cell: Produces energy by a spontaneous reaction which produces electricity as a result of electron transferred. Discharging of battery, corrosion, etc
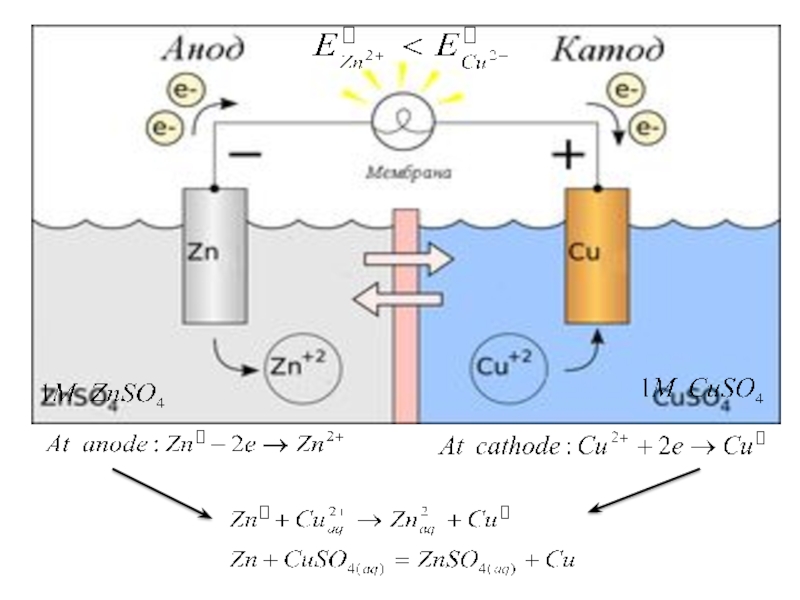
Слайд 28 Galvanic cell composed of two half-cells; which each consist of
a metal rod or strip immersed in a solution of
its own ions or an inert electrolyte.Electrodes: solid metal conductors connecting the cell to an external circuit.
Anode: electrode where oxidation occurs (-).
Cathode: electrode where reduction occurs (+).
The electrons flow from the anode to the cathode (“a before c”) through an electrical circuit rather than passing directly from one substance to another.
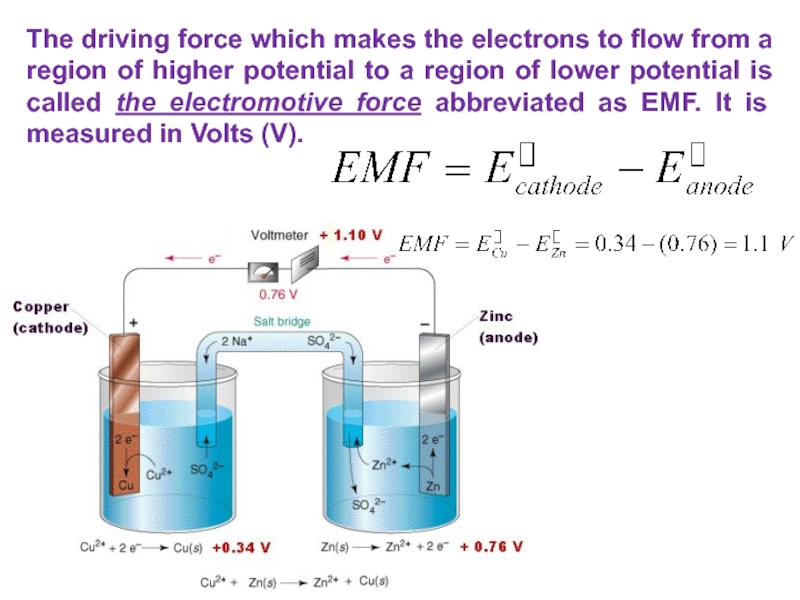
Слайд 29The driving force which makes the electrons to flow from
a region of higher potential to a region of lower
potential is called the electromotive force abbreviated as EMF. It is measured in Volts (V).Слайд 30If the E>0, it is positive, the reaction occurring is
spontaneous.
If the E
is non-spontaneousTHE ELECTROMOTIVE FORCE
Слайд 32Representation of a galvanic cell
Galvanic cell consists of two electrodes,
anode and cathode
The anode is written on the left hand
side while the cathode is written on right side.The anode is written with the metal first and then the electrolyte .The two are separated by a vertical line or semicolon. Example:
The cathode is written with electrolyte first and then the metal both are separated by vertical line or semicolon. Example:
The two half cells are connected by a salt bridge which is indicated by two parallel lines:
Слайд 33 Many natural phenomena are based on electrochemical processes, such as the corrosion
of metals, the ability of some sea creatures to generate
electrical fields, and the workings of the nervous systems of humans and other animals.They also play an important role in modern technology, most prominently in the storage of electrical power in batteries, and the electrochemical process called electrolysis is important in modern industry.
Слайд 34 Electric batteries use electrochemical processes to store and release electricity.
Chemical reactions within the electric cells making up the battery
create a difference in charge between the two halves of each cell, producing electrical current. Rechargeable batteries produce electricity with chemical reactions that are reversible, and so can be returned to their original chemical configuration if electricity is applied from an outside source. The reactions in nonrechargable batteries do not have this quality, though they usually produce more electric power than a rechargeable battery can provide in a single charge.A variety of different chemical reactions are used in batteries. Nickel-cadmium batteries, which are commonly used in lights and household appliances, are based on separate reactions of cadmium and nickel with an alkaline, usually a solution of potassium hydroxide (KOH), and water. Nickel-metal hydride batteries are similar, but replace the cadmium with an intermetallic compound made from manganese, aluminum, or cobalt mixed with rare earth metals such as praseodymium, lanthanum, and cerium.
Lithium batteries can use a variety of reactions involving lithium compounds, with the most common type using manganese dioxide (MnO2) and a solution of lithium perchlorate (LiClO4), dimethoxyethane (C4H10O2), and propylene carbonate (C4H6O3).