Разделы презентаций
- Разное
- Английский язык
- Астрономия
- Алгебра
- Биология
- География
- Геометрия
- Детские презентации
- Информатика
- История
- Литература
- Математика
- Медицина
- Менеджмент
- Музыка
- МХК
- Немецкий язык
- ОБЖ
- Обществознание
- Окружающий мир
- Педагогика
- Русский язык
- Технология
- Физика
- Философия
- Химия
- Шаблоны, картинки для презентаций
- Экология
- Экономика
- Юриспруденция
Electrochemistry
Содержание
- 1. Electrochemistry
- 2. Basic termselectric current molten stateto flow potentialcircuit electromotive force cell fuel cellelectrodesalt bridgesoluteanioncation
- 3. Electrochemistry and RedoxOxidation-reduction: “Redox”Electrochemistry: study of the interchange between
- 4. RedoxOxidation is loss of e- O.N. increases (more
- 5. Слайд 5
- 6. Слайд 6
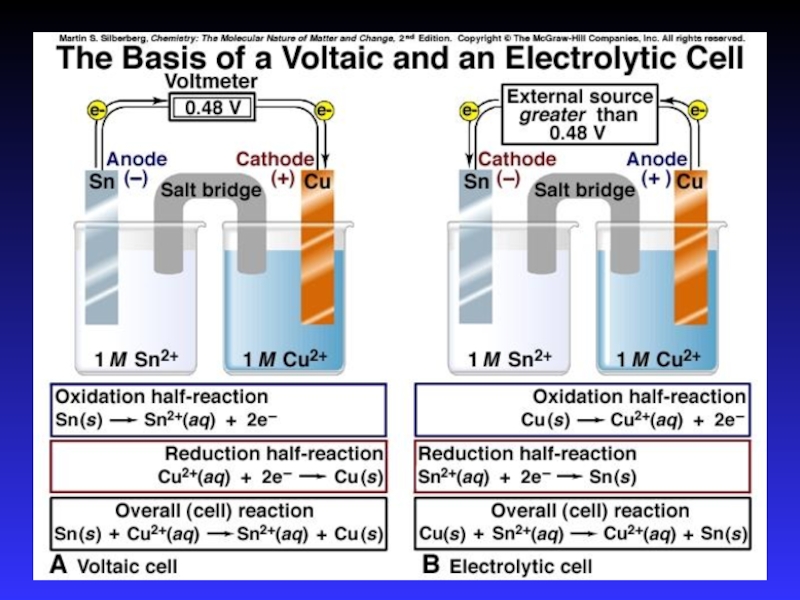
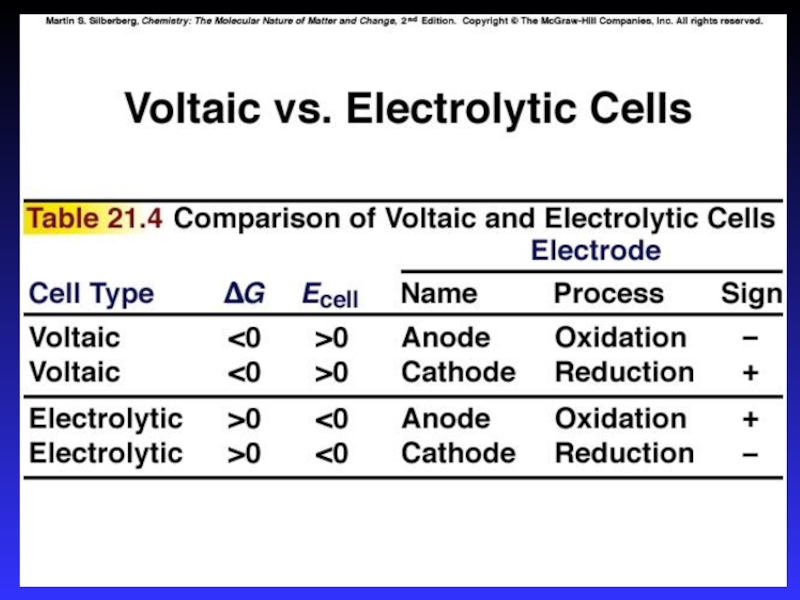
- 7. Types of cellsVoltaic (galvanic) cells:a spontaneous reaction
- 8. Common ComponentsElectrodes: conduct electricity between cell and surroundingsElectrolyte: mixture
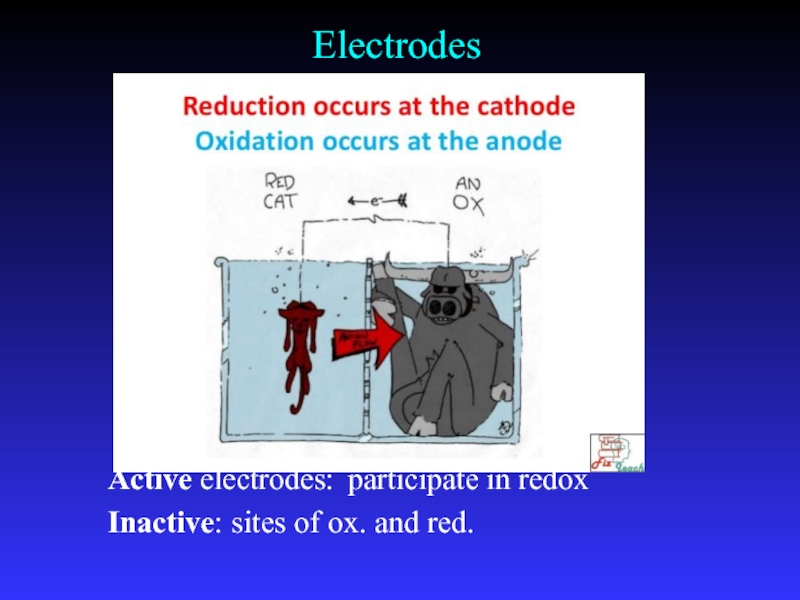
- 9. ElectrodesActive electrodes: participate in redoxInactive: sites of ox. and red.
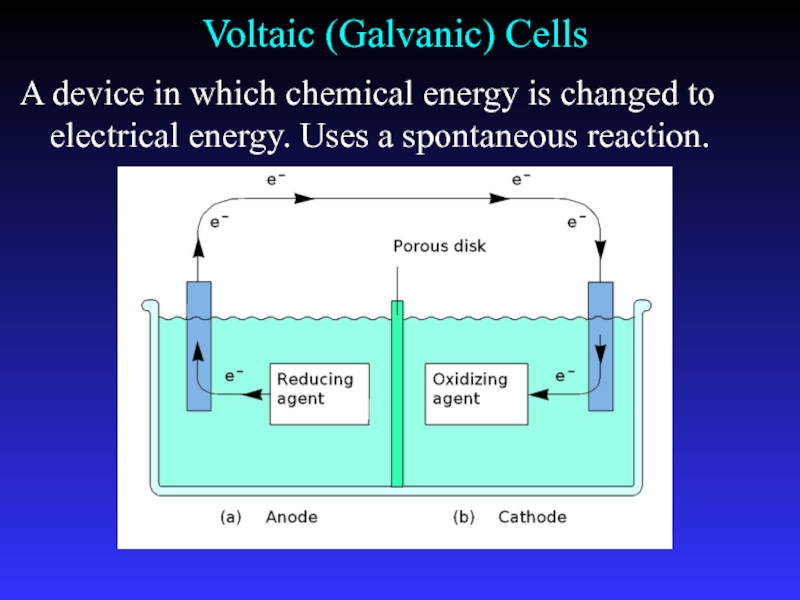
- 10. Voltaic (Galvanic) CellsA device in which chemical
- 11. Alessandro Volta (1745–1827) Luigi Galvani (1737-1798)
- 12. Слайд 12
- 13. Слайд 13
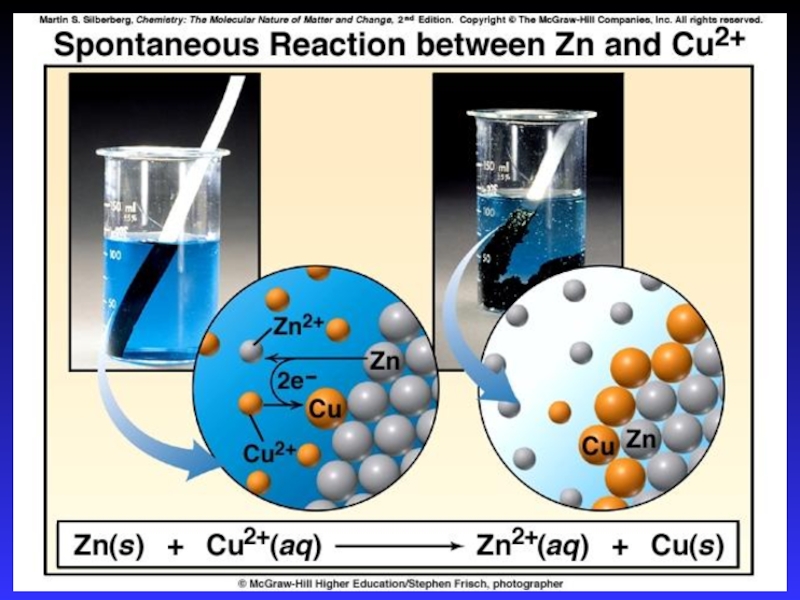
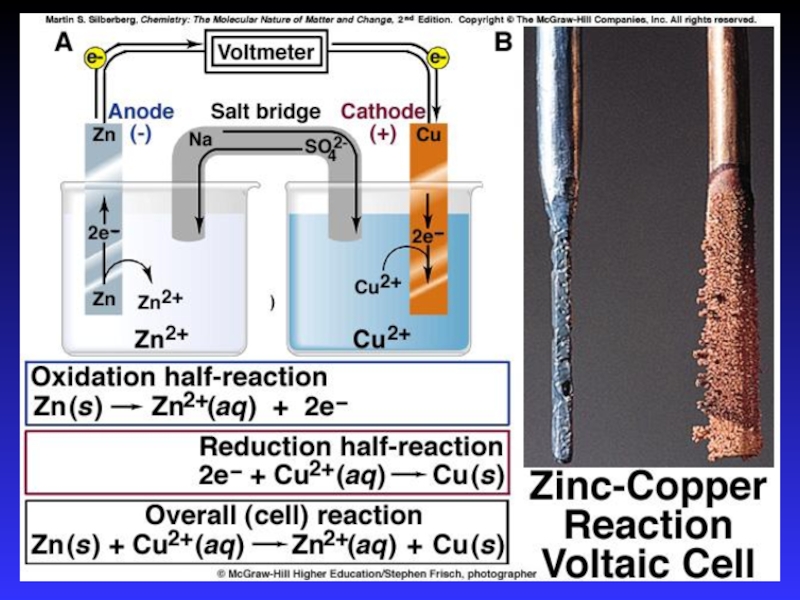
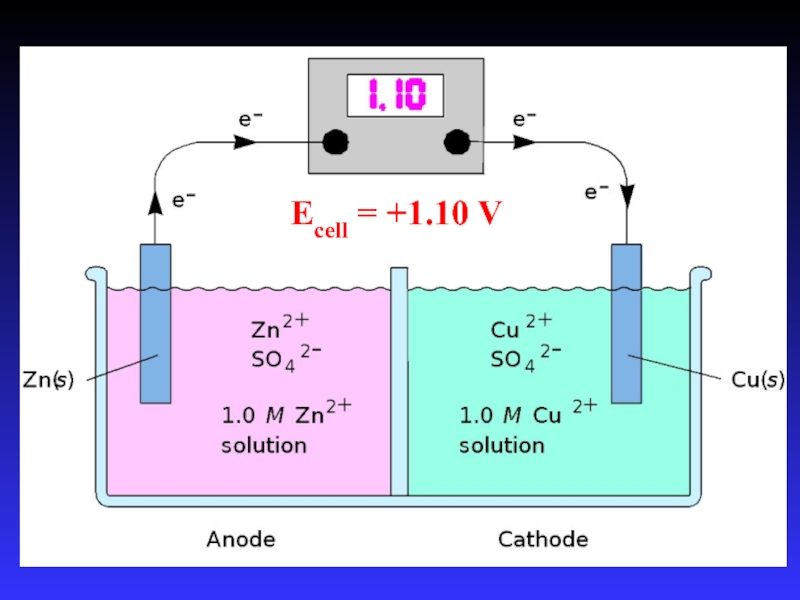
- 14. Zn2+(aq) + Cu(s) Cu2+(aq) + Zn(s)Zn
- 15. Designing a cell• half-equations representing reactions in
- 16. Ecell = +1.10 V
- 17. Слайд 17
- 18. A galvanic cell consists of an oxidizing
- 19. Standard Reduction PotentialsE0 values for reduction half-reactions
- 20. Слайд 20
- 21. Слайд 21
- 22. Calculating E0cellE0cell = E0cathode - E0anodeE0cell > 0 SpontaneousE0cell < 0 NotE0cell = 0 Equilibrium
- 23. Слайд 23
- 24. The Nernst equation is an equation that
- 25. Nernst EquationUnder nonstandard conditions
- 26. Ecell is the cell potential (electromotive force)
- 27. BatteriesA battery is a galvanic cell or,
- 28. Leclanché Acidic Dry CellElectrolyte in paste formZnCl2
- 29. Alkaline Dry CellSame basic cell as acidic
- 30. Lead Storage BatterySix cells in seriesElectrolyte =
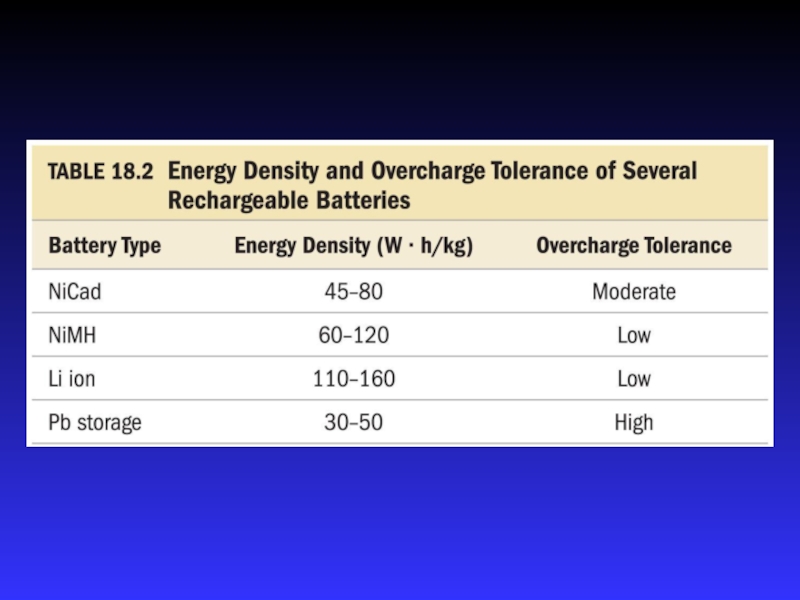
- 31. NiCad BatteryElectrolyte is concentrated KOH solutionAnode =
- 32. Ni-MH BatteryElectrolyte is concentrated KOH solutionAnode =
- 33. Lithium Ion BatteryElectrolyte is concentrated KOH solutionAnode
- 34. Слайд 34
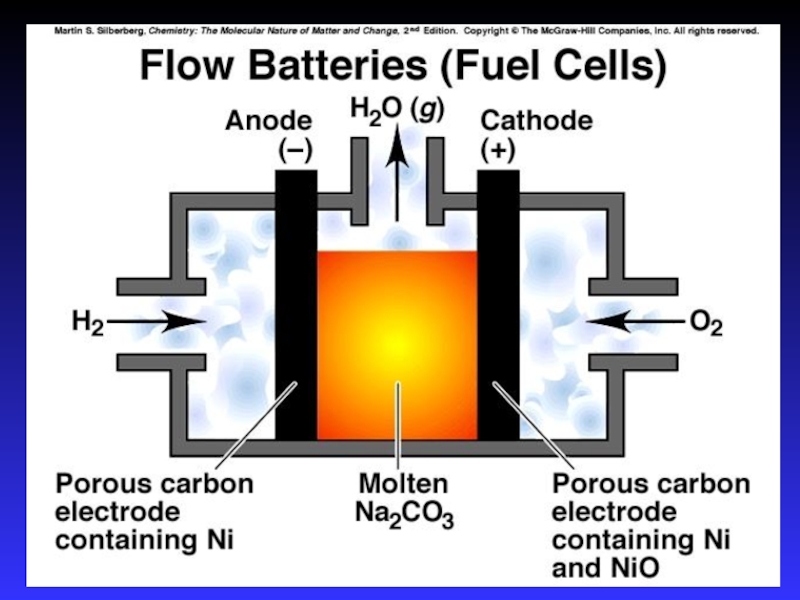
- 35. Fuel CellsLike batteries in which reactants are
- 36. Слайд 36
- 37. Electrolysis - the process of using electrical
- 38. Слайд 38
- 39. Слайд 39
- 40. Electrolytic CellsThe source of energy: a battery
- 41. Слайд 41
- 42. Слайд 42
- 43. Слайд 43
- 44. Michael Faraday (1791- 1867)1821 - discovered electromagnetic
- 45. Quantitative electrolysis and Faraday's laws
- 46. Слайд 46
- 47. Слайд 47
- 48. Home taskRead and memorize pp.333-335. (pp.302-339)Questions 1-11 p.336 24, 25 p.338(in writing)
- 49. Electrochemistry: Crash Course Chemistry #36
- 50. Скачать презентанцию
Basic termselectric current molten stateto flow potentialcircuit electromotive force cell fuel cellelectrodesalt bridgesoluteanioncation
Слайды и текст этой презентации
Слайд 2Basic terms
electric current molten state
to flow potential
circuit electromotive force
cell fuel cell
electrode
salt bridge
solute
anion
cation
Слайд 3Electrochemistry and Redox
Oxidation-reduction: “Redox”
Electrochemistry:
study of the interchange between chemical change and
electrical work
use electrical energyСлайд 4Redox
Oxidation is loss of e-
O.N. increases (more positive)
Reduction is gain
of e-
O.N. decreases (more negative)
Oxidation involves loss OIL
Reduction involves gain RIG
Слайд 7Types of cells
Voltaic (galvanic) cells:
a spontaneous reaction generates electrical energy
Chemistry→Electricity
Electrolytic
cells:
absorb free energy from an electrical source to drive a
nonspontaneous reactionElectricity→Chemistry
Слайд 8Common Components
Electrodes:
conduct electricity between cell and surroundings
Electrolyte:
mixture of ions involved
in reaction or carrying charge
Salt bridge:
completes circuit (provides charge balance)
Слайд 10Voltaic (Galvanic) Cells
A device in which chemical energy is changed
to electrical energy. Uses a spontaneous reaction.
Слайд 14Zn2+(aq) + Cu(s) Cu2+(aq) + Zn(s)
Zn gives up electrons
to Cu
“pushes harder” on e-
greater potential energy
greater “electrical potential”
Spontaneous
reaction due to relative difference in metals’ abilities to give e-
ability of e- to flow
Слайд 15Designing a cell
• half-equations representing reactions in each half-cell
• overall
ionic equation
• polarity of electrodes and their nature (anode and
cathode)• oxidizing agent and reducing agent
• direction of flow of electrons through the conducting wires and of ions
through the salt bridge
• physical changes occurring at the electrodes or in their vicinity (metal
deposition, electrode dissolution, changes in pH and so on).
Слайд 18A galvanic cell consists of an oxidizing agent (in cathode
half-cell) and a reducing agent (in anode half-cell).
Electrons flows through
a wire from the anode half-cell to the cathode half-cell.The driving force that allows electrons to flow is called the electromotive force (emf) or the cell potential (Ecell).
The unit of electrical potential is volt (V).
1 V = 1 J/C of charge transferred.
Cell Potential
Слайд 19Standard Reduction Potentials
E0 values for reduction half-reactions with solutes at
1M and gases at 1 atm
Cu2+ + 2e Cu
E0
= 0.34 V vs. SHESO42 + 4H+ + 2e H2SO3 + H2O
E0 = 0.20 V vs. SHE
Слайд 22Calculating E0cell
E0cell = E0cathode - E0anode
E0cell > 0 Spontaneous
E0cell
< 0 Not
E0cell = 0 Equilibrium
Слайд 24The Nernst equation is an equation that relates the reduction
potential of an electrochemical reaction (half-cell or full cell reaction)
to the standard electrode potential, temperature, and activities (often approximated by concentrations) of the chemical species undergoing reduction and oxidation.Walther Nernst
(1864-1941)
Слайд 26Ecell is the cell potential (electromotive force) at the temperature
of interest,
Eocell is the standard cell potential,
R is the
universal gas constant: R = 8.314472(15) J K−1 mol−1, T is the temperature in kelvins
F = 9.64853399(24)×104 C mol−1,
Qr is the reaction quotient of the cell reaction.
Слайд 27Batteries
A battery is a galvanic cell or, more commonly, a
group of galvanic cells connected in series.
Слайд 28Leclanché Acidic Dry Cell
Electrolyte in paste form
ZnCl2 + NH4Cl
Or MgBr2
Anode
= Zn (or Mg)
Zn(s) ® Zn2+(aq) + 2 e−
Cathode
= graphite rodMnO2 is reduced.
2 MnO2(s) + 2 NH4+(aq) + 2 H2O(l) + 2 e−
® 2 NH4OH(aq) + 2 Mn(O)OH(s)
Cell voltage = 1.5 V
Expensive, nonrechargeable, heavy, easily corroded
Слайд 29Alkaline Dry Cell
Same basic cell as acidic dry cell, except
electrolyte is alkaline
KOH paste
Anode = Zn (or Mg)
Zn(s) ®
Zn2+(aq) + 2 e− Cathode = graphite or brass rod
MnO2 is reduced.
2 MnO2(s) + 2 NH4+(aq) + 2 H2O(l) + 2 e−
® 2 NH4OH(aq) + 2 Mn(O)OH(s)
Cell voltage = 1.54 V
Longer shelf life than acidic dry cells and rechargeable, with little corrosion of zinc.
Слайд 30Lead Storage Battery
Six cells in series
Electrolyte = 30% H2SO4
Anode =
Pb
Pb(s) + SO42−(aq) ® PbSO4(s) + 2 e−
Cathode =
Pb coated with PbO2PbO2 is reduced.
PbO2(s) + 4 H+(aq) + SO42−(aq) + 2 e−
® PbSO4(s) + 2 H2O(l)
Cell voltage = 2.09 V
Rechargeable, heavy
Слайд 31NiCad Battery
Electrolyte is concentrated KOH solution
Anode = Cd
Cd(s) + 2
OH−(aq) → Cd(OH)2(s) + 2 e−
E0 = 0.81 V
Cathode
= Ni coated with NiO2NiO2 is reduced.
NiO2(s) + 2 H2O(l) + 2 e− → Ni(OH)2(s) + 2OH− E0 = 0.49 V
Cell voltage = 1.30 V
Rechargeable,
long life, light;
however, recharging incorrectly
can lead to battery breakdown
Слайд 32Ni-MH Battery
Electrolyte is concentrated KOH solution
Anode = metal alloy with
dissolved hydrogen
Oxidation of H from H0 to H+
M ∙ H(s)
+ OH−(aq) → M(s) + H2O(l) + e− E° = 0.89 V
Cathode = Ni coated with NiO2
NiO2 is reduced.
NiO2(s) + 2 H2O(l) + 2 e− → Ni(OH)2(s) + 2OH−
E0 = 0.49 V
Cell voltage = 1.30 V
Rechargeable, long life, light, more environmentally friendly than NiCad, greater energy density than NiCad
Слайд 33Lithium Ion Battery
Electrolyte is concentrated KOH solution
Anode = graphite impregnated
with Li ions
Cathode = Li - transition
metal oxide
Reduction of
transition metalWork on Li ion migration from anode to cathode causing a corresponding migration of electrons from anode to cathode
Rechargeable, long life, very light, more environmentally friendly, greater energy density
Слайд 35Fuel Cells
Like batteries in which reactants are constantly being added
So
it never runs down!
Anode and cathode both Pt
coated metal
Electrolyte
is OH– solution.Anode reaction
2 H2 + 4 OH– → 4 H2O(l) + 4 e−
Cathode reaction
O2 + 4 H2O + 4 e− → 4 OH–
Слайд 37Electrolysis
- the process of using electrical energy to break
a compound apart.
Electrolysis is done in an electrolytic cell.
Electrolytic cells
can be used to separate elements from their compounds.Слайд 40Electrolytic Cells
The source of energy: a battery or DC power
supply.
The positive terminal of the source is attached to the
anode.The negative terminal of the source is attached to the cathode.
Electrolyte can be either an aqueous salt solution or a molten ionic salt.
Cations in the electrolyte are attracted to the cathode and anions are attracted to the anode.
Cations pick up electrons from the cathode and are reduced; anions release electrons to the anode and are oxidized.
Слайд 44Michael Faraday (1791- 1867)
1821 - discovered electromagnetic rotation.
1831 -
discovered electromagnetic induction, the principle behind the electricity generator.
1825
- isolated benzene. 1830 - became professor of chemistry at the Royal Military Academy in Woolwich
1834 - laws of electrolysis