Слайд 1An Uncommon Brainstem Lesion in a Young Patient
Aleksandra Parfenova
PGY3
First Saint-Petersburg Pavlov State Medical University
Institute of Human Brain
of Russian Academy of Sciences (IHB RAS) n.a. N.P.Bechtereva
September, 8th, 2016
Слайд 2Case
20 YO male
Admitted to the Neurology department on April, 11th,
2016
Complaining of gait ataxia and speech difficulty.
PMH:
No previous illnesses, no
trauma
Medications – none
Allergies – none
Social History - Non-smoker, no alcohol intake, no drugs
Family History – None
Nationality – Latvian
Living with his father, studying at home
Слайд 3History of present illness
2010 – sudden vertigo, horizontal diplopia, oculomotor
abnormalities, gait ataxia. He was transferred to the hospital
Neurological symptoms
resolved with pulse methylprednisolone.
One week later, the same symptoms recurred and did not respond to pulse steroid. Symptoms persisted.
During the hospitalization in 2010, he developed recurrent episodes of right-sided weakness lasting ~5min each time.
Over the next 3 years, he had 2 more exacerbations with gait ataxia. Methylprednisolone infusions and recurrent plasma exchanges were not effective.
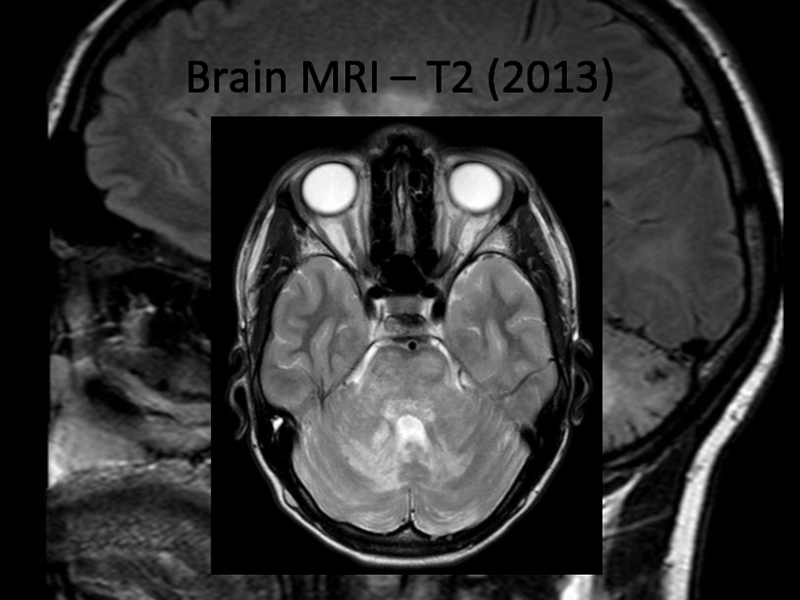
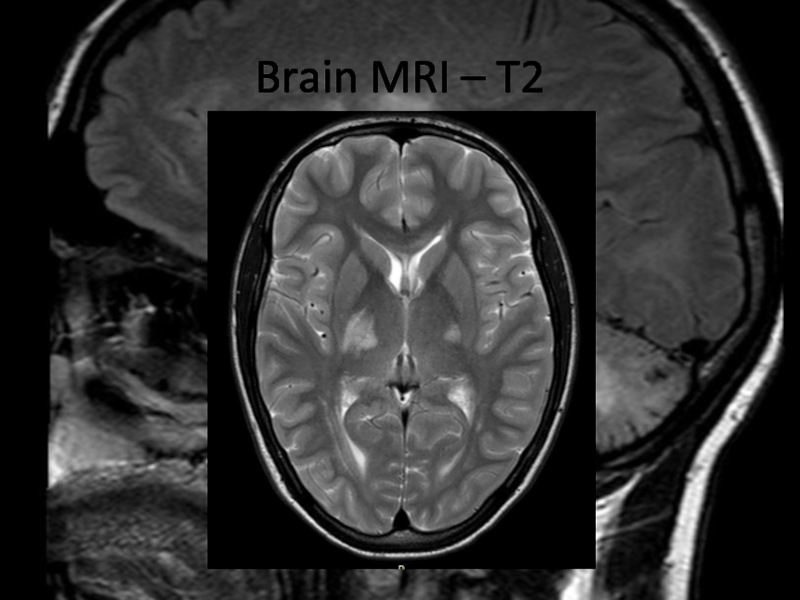
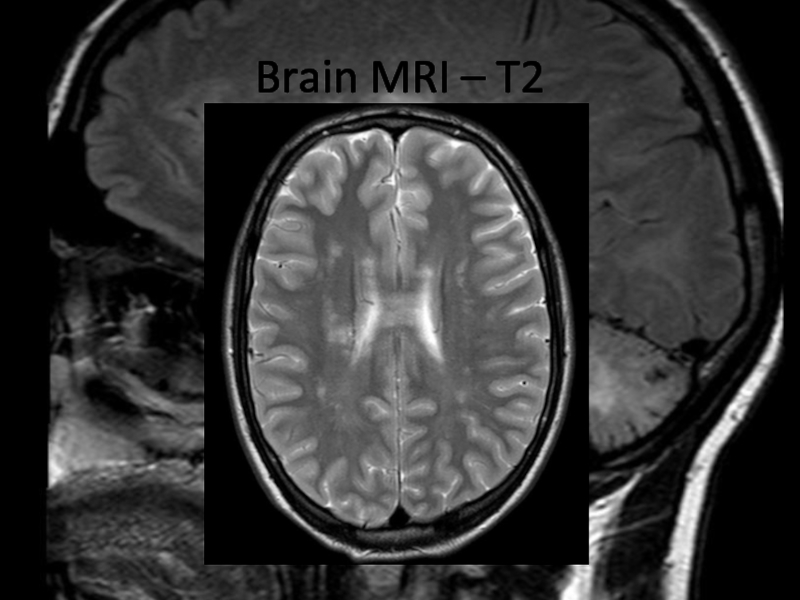
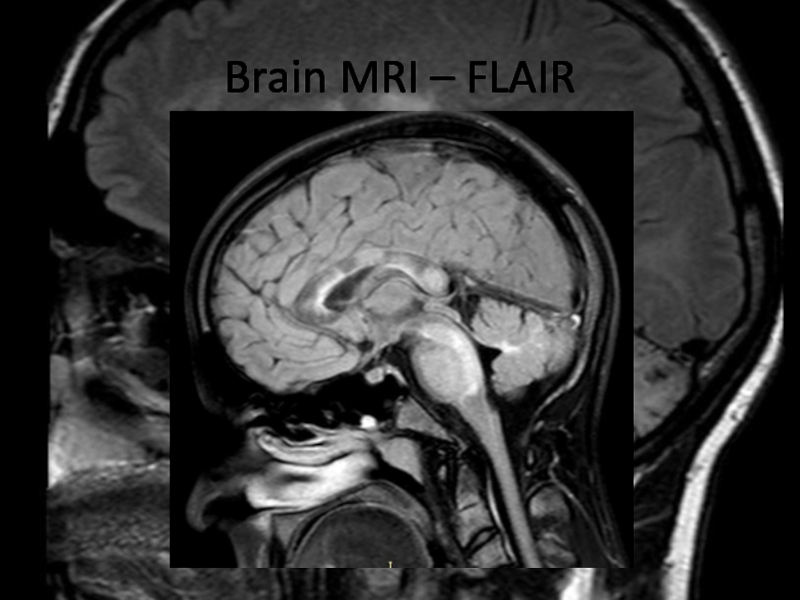
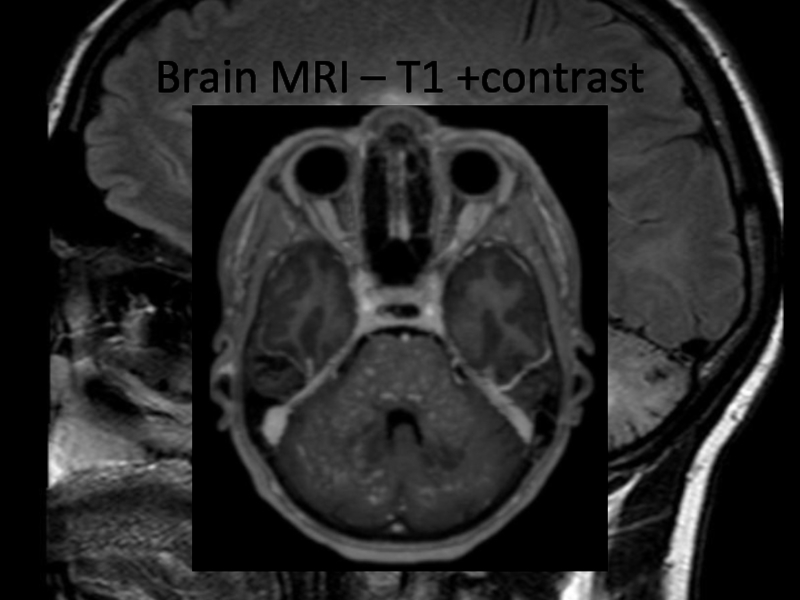
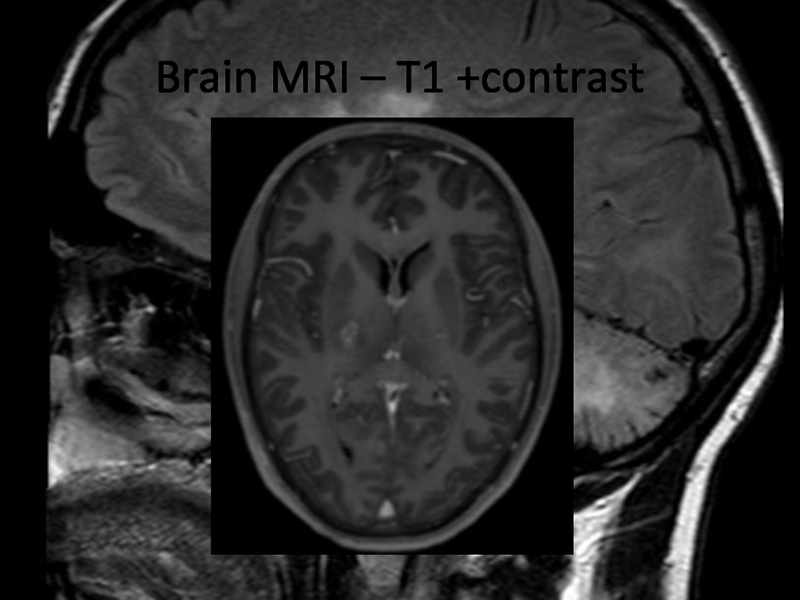
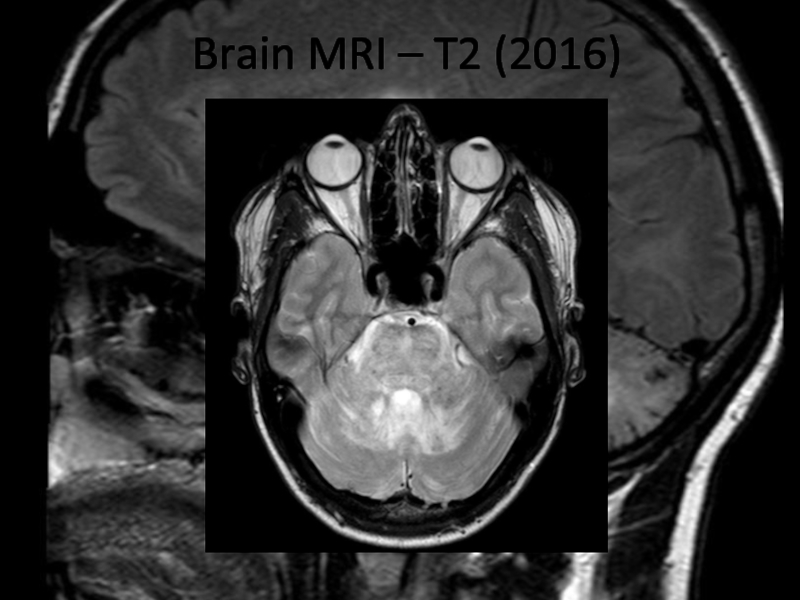
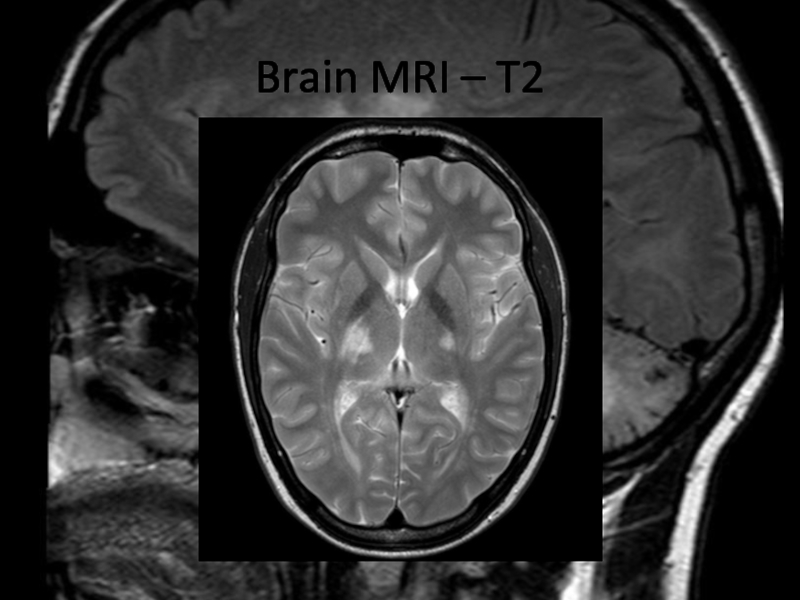
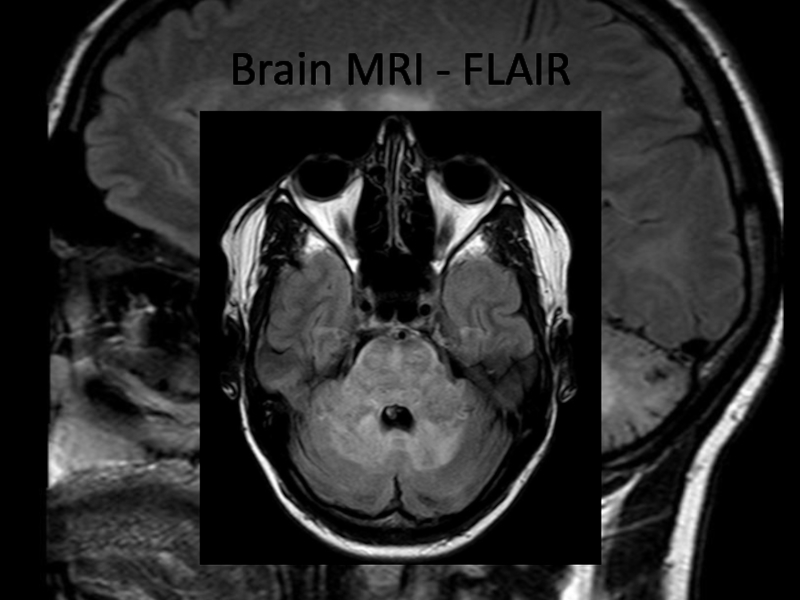
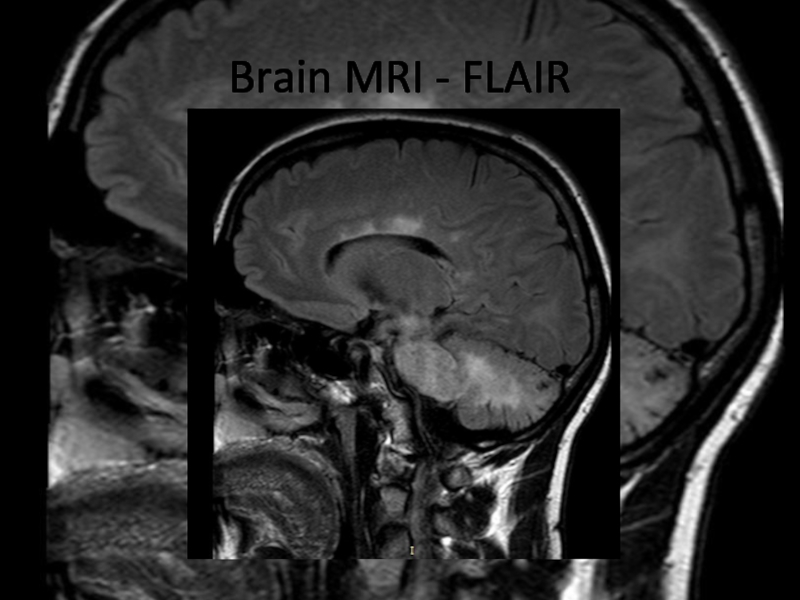
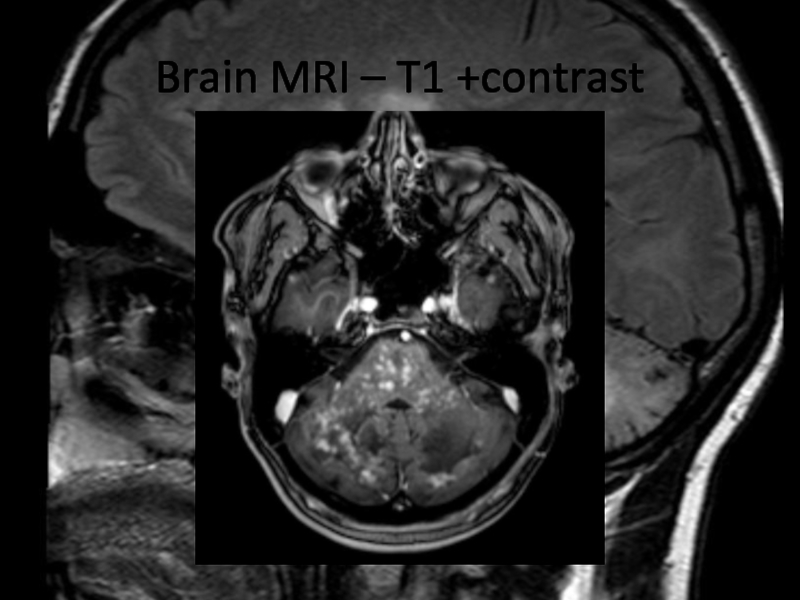
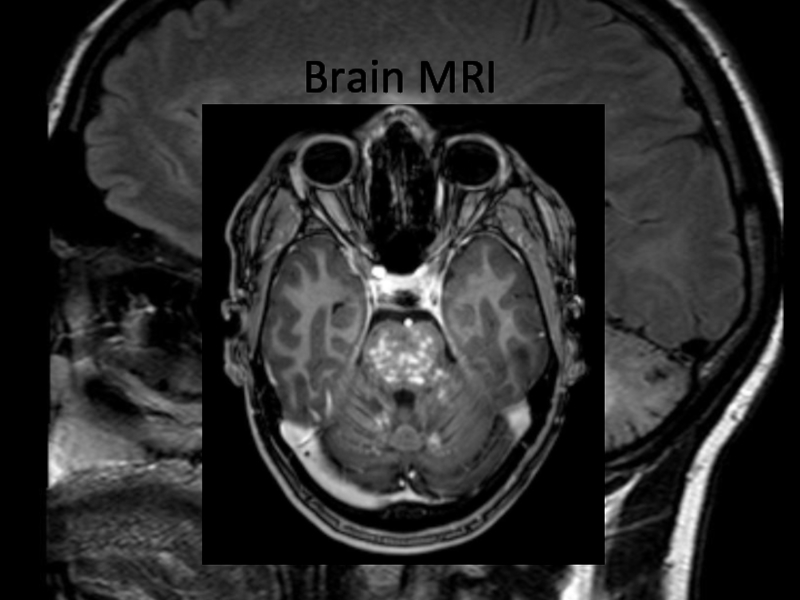
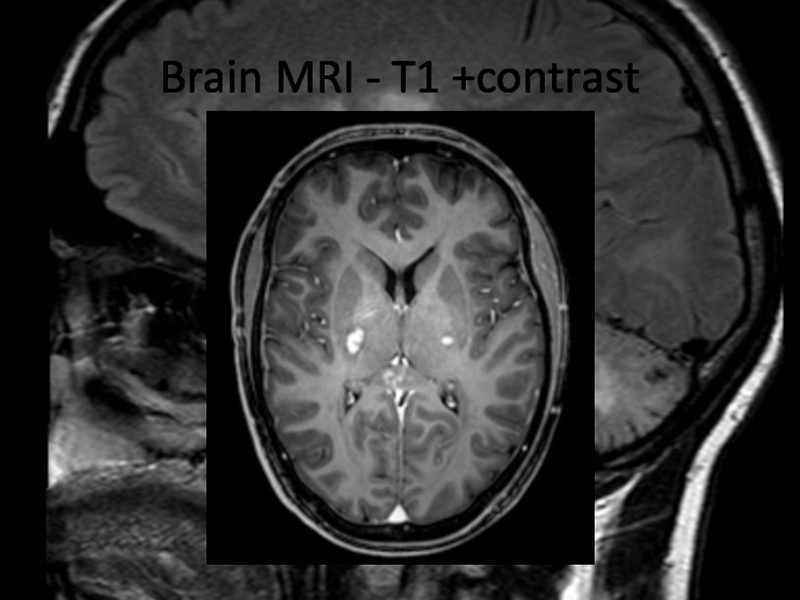
In 2013 brain MRI was done.
Слайд 10questions
Describe the findings of the MRI scans.
What is the
differential diagnosis?
What investigations would you pursue?
What treatment would you
propose?
Слайд 11Differential diagnosis
MS
ADEM
Neurosarcoidosis,
Sjögren's syndrome
NMO
Autoimmune encephalitides
CNS vasculitis
Primary angiitis of
CNS
CNS infections
Lymphoma
Glioma
Paraneoplastic syndromes
Final diagnosis (2013): Cerebral vasculitis
Слайд 12Treatment during the next years:
May 2014: planned hospitalization to
start prophylactic therapy with prednisolone 15 mg per day x
2 months, and methotrexate 10 mg once a week.
In March 2015: new exacerbation with increase of gait ataxia. Treated with pulse Methylprednisolone X 3days and increased prednisolone 30 mg/d X 3 months, methotrexate 10 mg once a week with positive effects.
September, 2nd, 2015: started i.v. Rituximab 1000 mg
But in a week, recurrent exacerbation developed also with further increase of gait ataxia. Treated with pulse Methylprednisolone with good effects followed by oral Prednisolone 30 mg/d x 4 months.
Each Prednisolone withdrawal led to deterioration of patient’s clinical status.
Слайд 13Examination
BP=120/80 mm Hg, Ps=68/min
Alert, oriented, normal higher cortical functions.
CN: Horizontal
nystagmus in left gaze. Vertical nystagmus in upgaze. Left lower
facial weakness. Dysarthria (bulbar syndrome).
Normal muscle strength in limbs, neck, trunk.
Reflexes: hyperreflexic, ankle clonus.
Bilateral upping Babinski's sign.
No sensory abnormalities.
Coordination tests – bilateral mild intention tremor. Bilateral dysdiadochokinesia. Severe gait ataxia.
Слайд 14Laboratory tests
CBA: Leuc.- 4.2 x10*9/l; er 5.18x10*12/l; Hb 159 g/l;
lymf. 29%; mon. 9%; neutr. 58%; eos. 4%;bas. 0%; SR
3 mm/h.
Biochemical BA: protein total 68 g/l; creatinine 60 mkmol/l; bilirubin total 10,1 mkmol/l; glucose 5.6 mmol/l.
Urine analysis: unit weight 1.025, protein 0 g/l; leuc. 0-0-1 in field of view; mucus +.
RW, HIV- negative.
Слайд 15Laboratory tests
Autoantibodies and markers of vasculitis: anti-nuclear antibodies (ANA), extractable
nuclear antigens (ENA), anti-neutrophil cytoplasmic antibodies (ANCA), HUVEC, aquaporin-4 antibodies
in blood - negative.
Oligoclonal IgG in serum and CSF (from the history of present illness) - negative.
Biochemical CSF test – no data
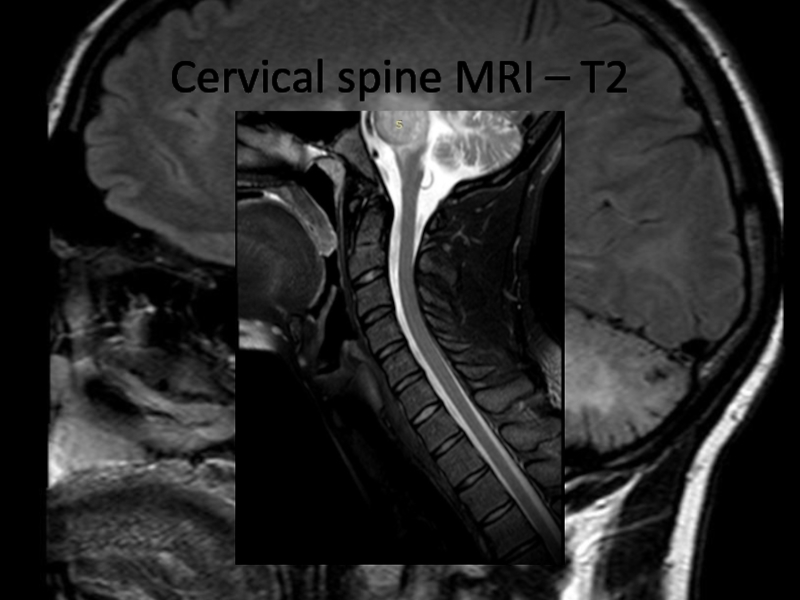
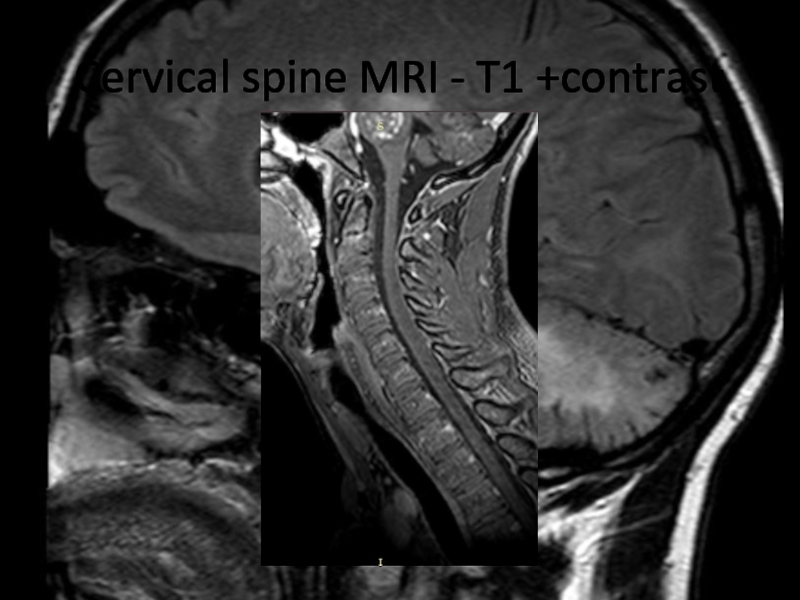
Слайд 24Cervical spine MRI - T1 +contrast
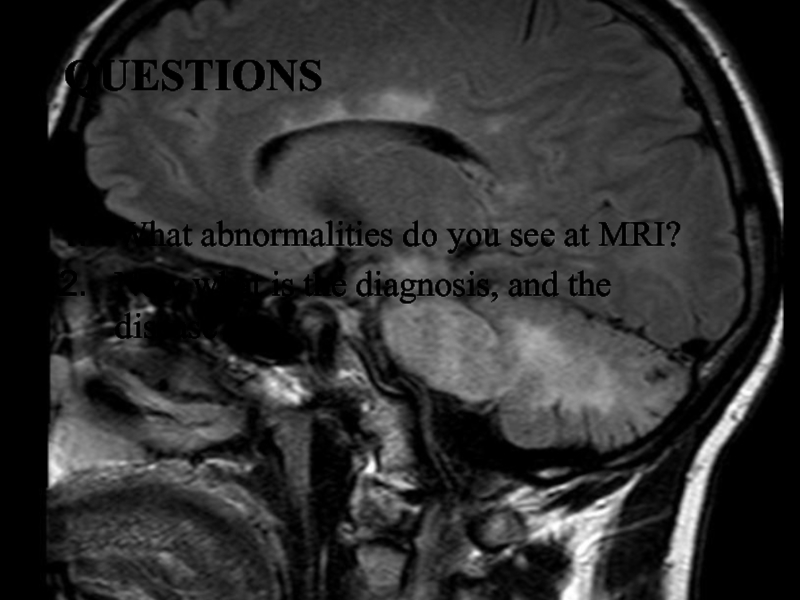
Слайд 25Questions
What abnormalities do you see at MRI?
Now what
is the diagnosis, and the disease?
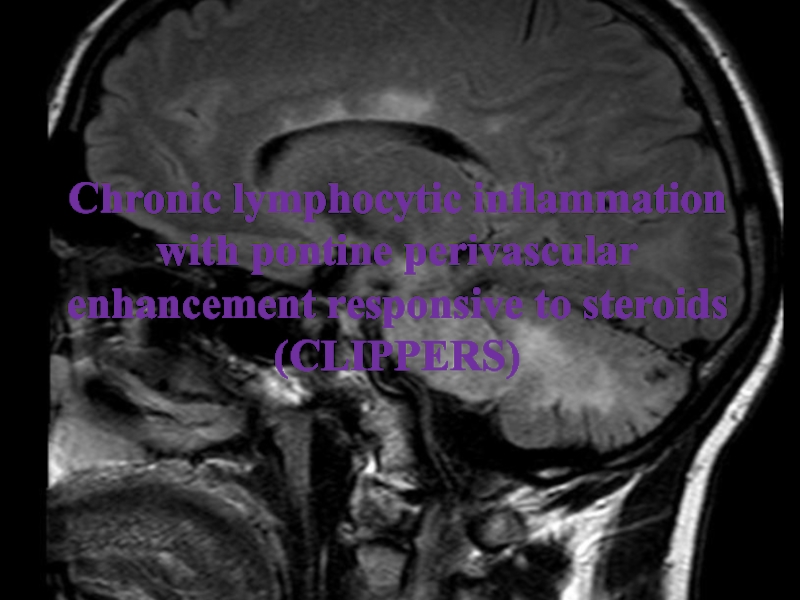
Слайд 26Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids
(CLIPPERS)
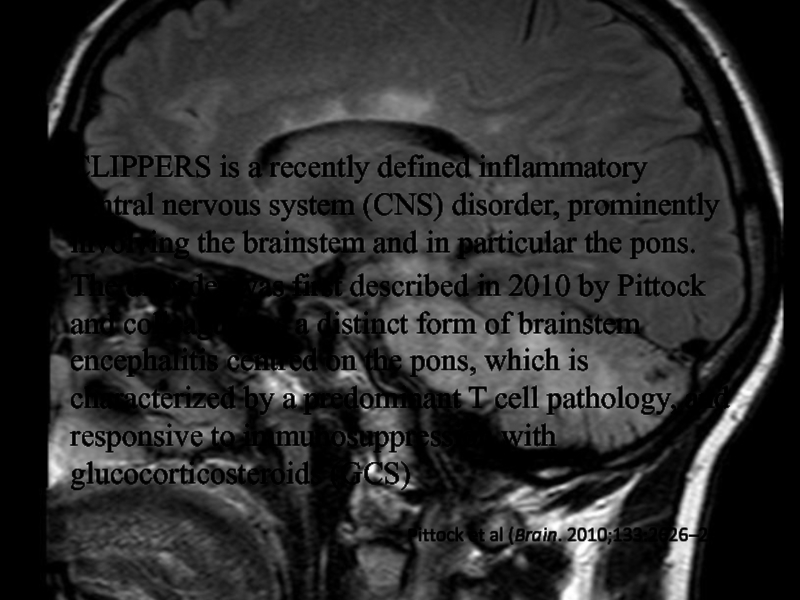
Слайд 27CLIPPERS is a recently defined inflammatory central nervous system (CNS)
disorder, prominently involving the brainstem and in particular the pons.
The
disorder was first described in 2010 by Pittock and colleagues as a distinct form of brainstem encephalitis centred on the pons, which is characterized by a predominant T cell pathology, and responsive to immunosuppression with glucocorticosteroids (GCS)
Pittock et al (Brain. 2010;133:2626–2634)
Слайд 28Pathogenesis
The pathogenesis of CLIPPERS is poorly understood and ultimately unknown.
The perivascular and T cell-predominant inflammatory cell infiltrates in affected
CNS lesions, patterns of CSF changes and typical gadolinium enhancement together with the clinico-radiological response to GCS-based immunosuppressive therapies suggest an (auto-)immune-mediated or other inflammatory pathogenesis.
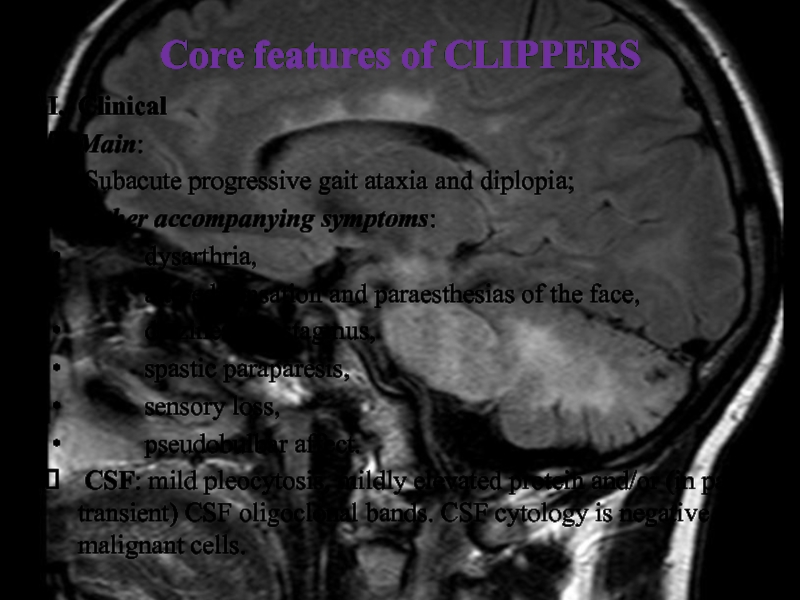
Слайд 29Core features of CLIPPERS
I. Clinical
Main:
Subacute progressive gait ataxia and diplopia;
Other accompanying symptoms:
dysarthria,
altered sensation and paraesthesias of the face,
dizziness, nystagmus,
spastic paraparesis,
sensory loss,
pseudobulbar affect.
CSF: mild pleocytosis, mildly elevated protein and/or (in part transient) CSF oligoclonal bands. CSF cytology is negative for malignant cells.
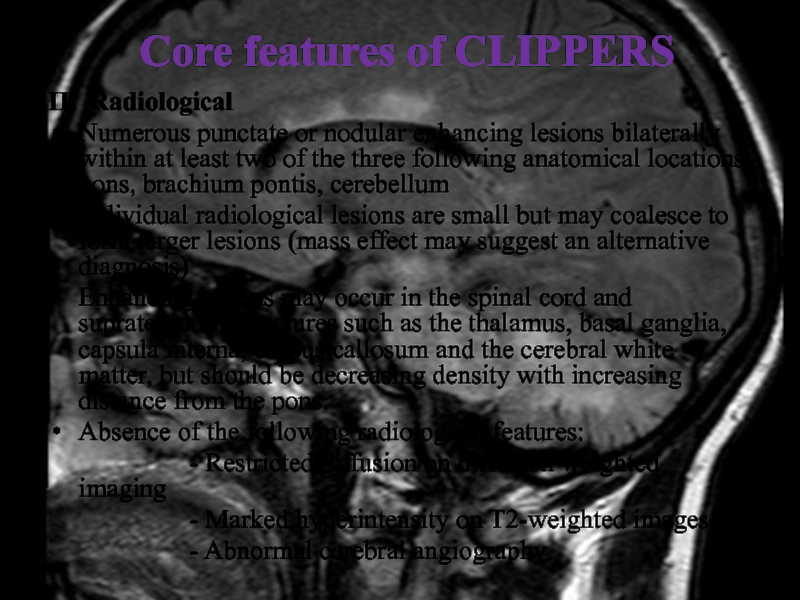
Слайд 30Core features of CLIPPERS
II. Radiological
Numerous punctate or nodular enhancing lesions bilaterally
within at least two of the three following anatomical locations:
pons, brachium pontis, cerebellum
Individual radiological lesions are small but may coalesce to form larger lesions (mass effect may suggest an alternative diagnosis)
Enhancing lesions may occur in the spinal cord and supratentorial structures such as the thalamus, basal ganglia, capsula interna, corpus callosum and the cerebral white matter, but should be decreasing density with increasing distance from the pons.
Absence of the following radiological features:
- Restricted diffusion on diffusion weighted imaging
- Marked hyperintensity on T2-weighted images
- Abnormal cerebral angiography
Слайд 33Core features of CLIPPERS
III. Glucocorticosteroid responsiveness
Clinical and radiological responsiveness to glucocorticosteroid
(GCS)-based immunosuppression.
However, the patients routinely worsened following GCS taper and
required chronic GCS or other immunosuppressive treatment as maintenance therapy.
Слайд 34Core features of CLIPPERS
IV. Histopathological
White matter perivascular lymphohistiocytic infiltrate with or
without parenchymal extension
Infiltrate contains predominantly CD3 and CD4 lymphocytes
Absence of
the following histopathological characteristics:
- Monoclonal or atypical lymphocyte population
- Necrotizing granulomas or giant cells
- Histological features of vasculitis
Слайд 35Core features of CLIPPERS
Differential diagnoses should be excluded e.g. neurosarcoidosis,
Sjögren's syndrome, neuro-Behçet's disease, MS, ADEM, NMO, Bickerstaff encephalitis, other
autoimmune encephalitides, CNS vasculitis, primary angiitis of CNS, CNS infections, histiocytosis, lymphoma, glioma, paraneoplastic syndromes
Слайд 36«Red flags»
No response to treatment with GCS at the beginning
or during follow-up.
Unusual clinical findings such as fever, extracerebral
organ manifestations (such as arthritis, uveitis, lymphadenopathy, etc.) and meningism should lead to increased alertness.
Dysarthria and ataxia are so common in CLIPPERS that their absence should be considered a hint that the disorder might be something else
MRI findings: although they may be subtle, abnormalities of brainstem are so common in CLIPPERS that their absence is worth noting. Pontine lesions with necrosis may point to a PCNSL and marked mass effects to CNS tumours in general
CSF findings: marked pleocytosis (> 100/μl) or malignant cells should prompt reevaluation of the diagnosis
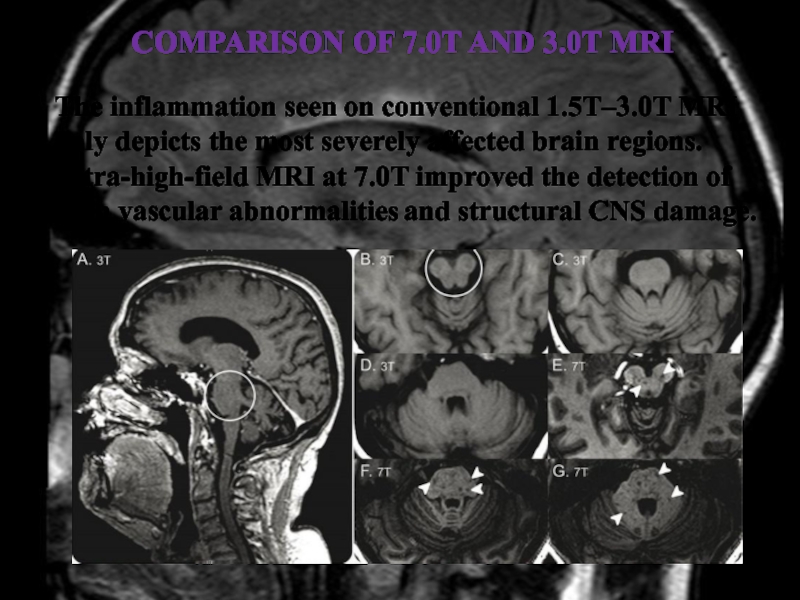
Слайд 37Comparison of 7.0T and 3.0T MRI
The inflammation seen on conventional
1.5T–3.0T MRI only depicts the most severely affected brain regions.
Ultra-high-field MRI at 7.0T improved the detection of both vascular abnormalities and structural CNS damage.
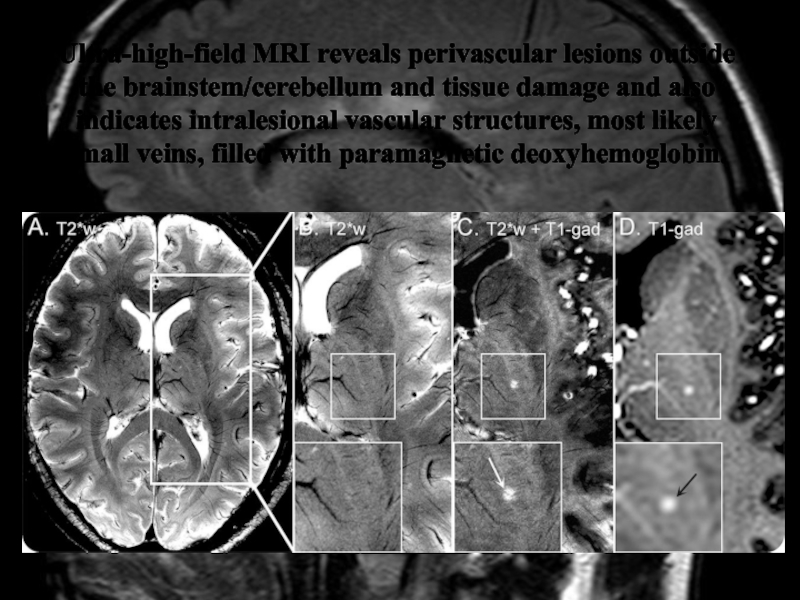
Слайд 38Ultra-high-field MRI reveals perivascular lesions outside the brainstem/cerebellum and tissue
damage and also indicates intralesional vascular structures, most likely small
veins, filled with paramagnetic deoxyhemoglobin.
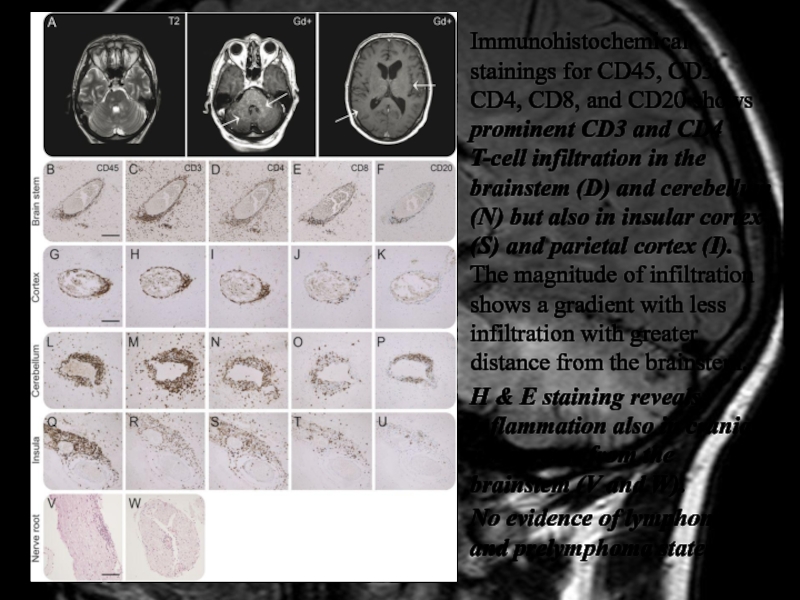
Слайд 39Immunohistochemical stainings for CD45, CD3, CD4, CD8, and CD20 shows
prominent CD3 and CD4 T-cell infiltration in the brainstem (D)
and cerebellum (N) but also in insular cortex (S) and parietal cortex (I). The magnitude of infiltration shows a gradient with less infiltration with greater distance from the brainstem.
H & E staining reveals inflammation also in cranial nerve roots from the brainstem (V and W).
No evidence of lymphoma and prelymphoma state.
Слайд 40Treatment
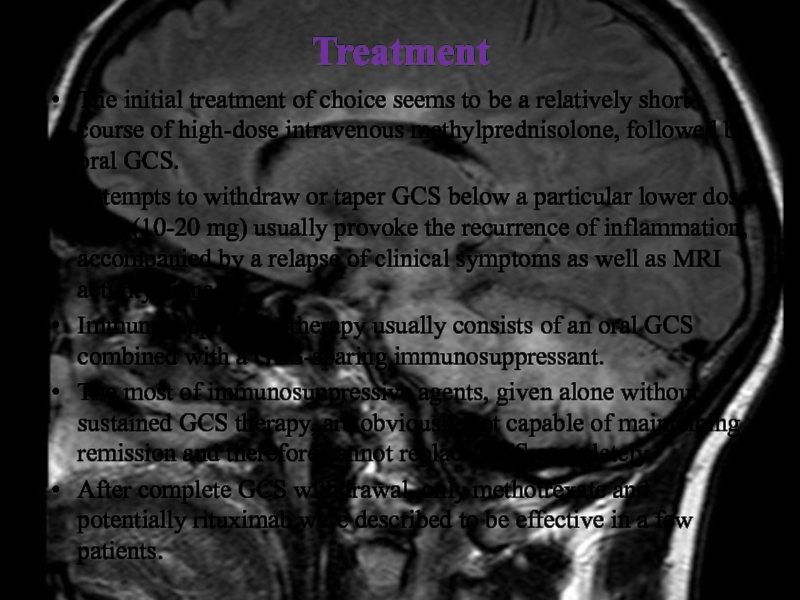
The initial treatment of choice seems to be a relatively
short course of high-dose intravenous methylprednisolone, followed by oral GCS.
Attempts to withdraw or taper GCS below a particular lower dose limit (10-20 mg) usually provoke the recurrence of inflammation, accompanied by a relapse of clinical symptoms as well as MRI activity signs.
Immunosuppressive therapy usually consists of an oral GCS combined with a GCS-sparing immunosuppressant.
The most of immunosuppressive agents, given alone without sustained GCS therapy, are obviously not capable of maintaining remission and therefore cannot replace GCS completely.
After complete GCS withdrawal, only methotrexate and potentially rituximab were described to be effective in a few patients.
Слайд 41Back to the case
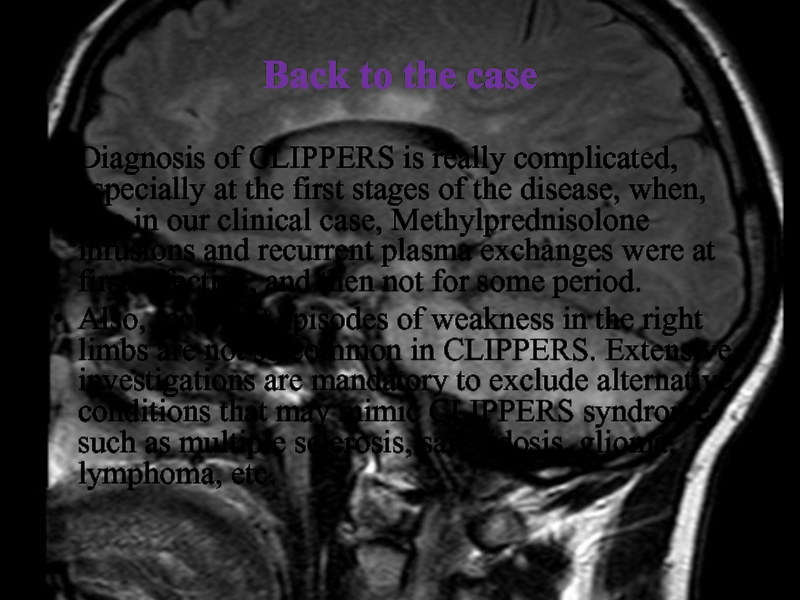
Diagnosis of CLIPPERS is really complicated, especially
at the first stages of the disease, when, like in
our clinical case, Methylprednisolone infusions and recurrent plasma exchanges were at first effective, and then not for some period.
Also, recurrent episodes of weakness in the right limbs are not so common in CLIPPERS. Extensive investigations are mandatory to exclude alternative conditions that may mimic CLIPPERS syndrome, such as multiple sclerosis, sarcoidosis, glioma, lymphoma, etc.
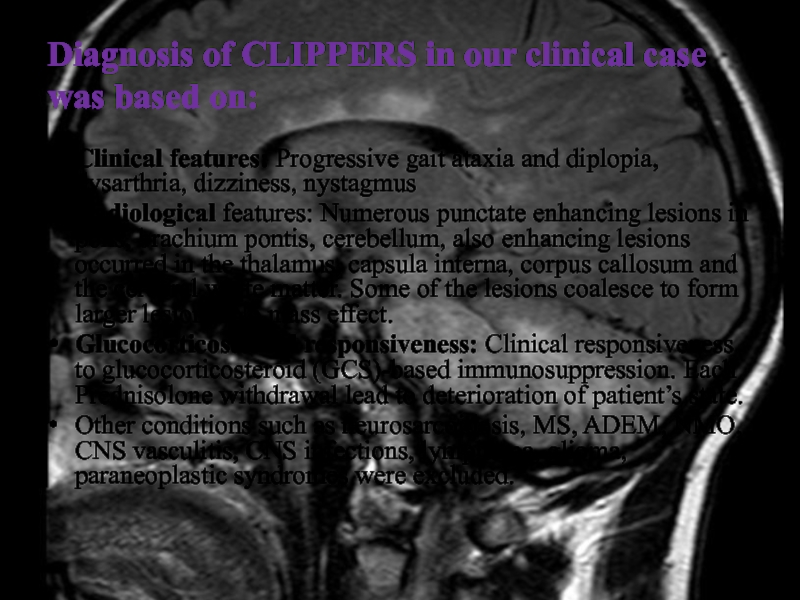
Слайд 42Diagnosis of CLIPPERS in our clinical case was based on:
Clinical
features: Progressive gait ataxia and diplopia, dysarthria, dizziness, nystagmus
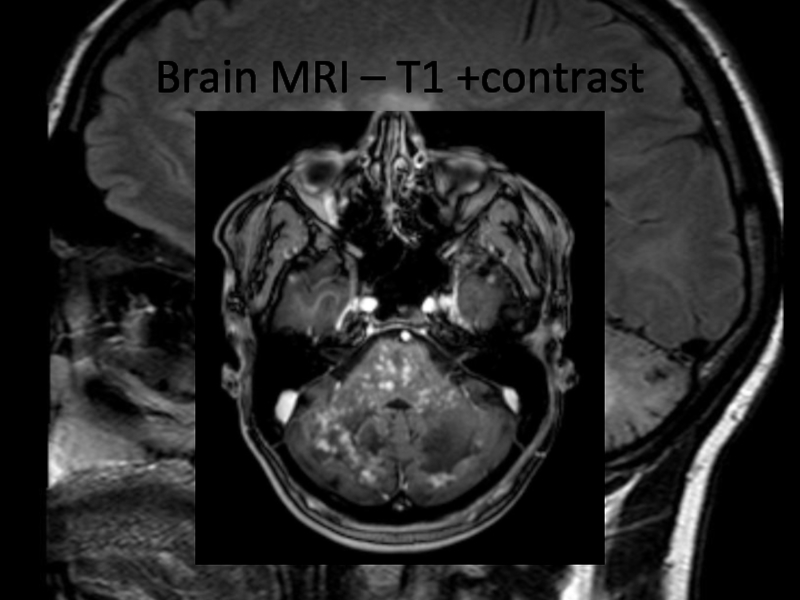
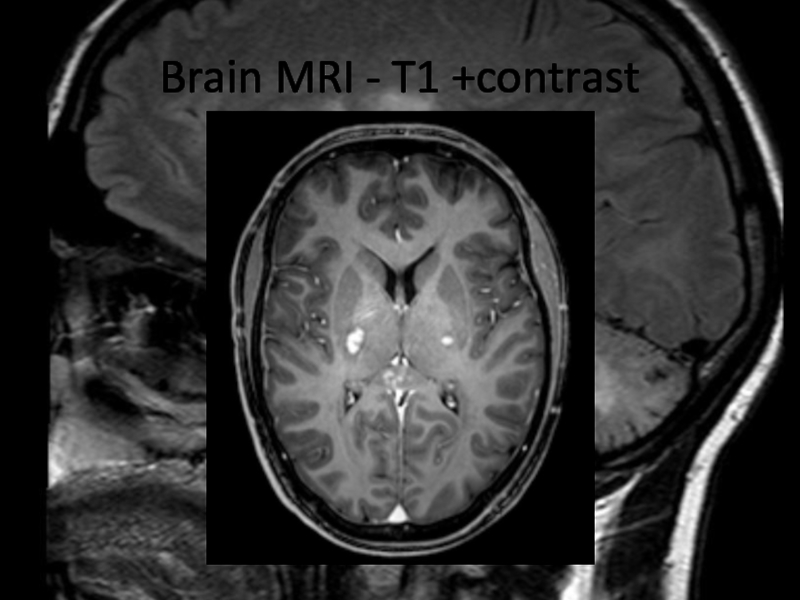
Radiological features:
Numerous punctate enhancing lesions in pons, brachium pontis, cerebellum, also enhancing lesions occurred in the thalamus, capsula interna, corpus callosum and the cerebral white matter. Some of the lesions coalesce to form larger lesions. No mass effect.
Glucocorticosteroid responsiveness: Clinical responsiveness to glucocorticosteroid (GCS)-based immunosuppression. Each Prednisolone withdrawal lead to deterioration of patient’s state.
Other conditions such as neurosarcoidosis, MS, ADEM, NMO, CNS vasculitis, CNS infections, lymphoma, glioma, paraneoplastic syndromes were excluded.
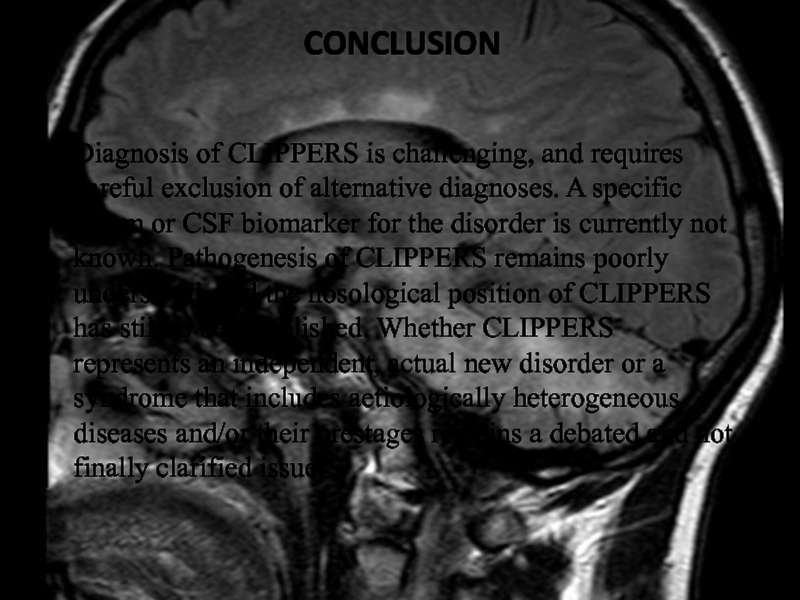
Слайд 43Conclusion
Diagnosis of CLIPPERS is challenging, and requires careful exclusion
of alternative diagnoses. A specific serum or CSF biomarker for
the disorder is currently not known. Pathogenesis of CLIPPERS remains poorly understood, and the nosological position of CLIPPERS has still to be established. Whether CLIPPERS represents an independent, actual new disorder or a syndrome that includes aetiologically heterogeneous diseases and/or their prestages remains a debated and not finally clarified issue.
Слайд 44References:
Buttmann M, Metz I, Brecht I, Brück W, Warmuth-Metz
M. Atypical chronic lymphocytic inflammation with pontocerebellar perivascular enhancement responsive
to steroids (CLIPPERS), primary angiitis of the CNS mimicking CLIPPERS or overlap syndrome? A case report. J Neurol Sci 2013;324:183–186.
Ferreira RM, Machado G, Souza AS, Lin K, Corrêa-Neto Y. CLIPPERS-like MRI findings in a patient with multiple sclerosis. J Neurol Sci 2013;327:61–62.
De Graaff HJ, Wattjes MP, Rozemuller-Kwakkel AJ, Petzold A, Killestein J. Fatal B-cell lymphoma following chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids. JAMA Neurol 2013;70:915–918.
Lin AW, Das S, Fraser JA, et al. Emergence of primary CNS lymphoma in a patient with findings of CLIPPERS. Can J Neurol Sci 2014;41:528–529.
Taieb G, Uro-Coste E, Clanet M, et al. A central nervous system B-cell lymphoma arising two years after initial diagnosis of CLIPPERS. J Neurol Sci 2014;344:224–226.
Müller K, Kuchling J, Dörr J, Harms L, Ruprecht K, Niendorf T. Detailing intra-lesional venous lumen shrinking in multiple sclerosis investigated by sFLAIR MRI at 7-T. J Neurol 2014;261:2032–2036.
Sinnecker T, Bozin I, Dörr J, et al. Periventricular venous density in multiple sclerosis is inversely associated with T2 lesion count: a 7 Tesla MRI study. Mult Scler 2013;19:316–325.
Слайд 45References:
Tallantyre EC, Brookes MJ, Dixon JE, Morgan PS, Evangelou N,
Morris PG. Demonstrating the perivascular distribution of MS lesions in
vivo with 7-Tesla MRI. Neurology 2008;70:2076–2078.
Kollia K, Maderwald S, Putzki N, et al. First clinical study on ultra-high-field MR imaging in patients with multiple sclerosis: comparison of 1.5T and 7T. AJNR Am J Neuroradiol 2009;30:699–702.
Gaitán MI, de Alwis MP, Sati P, Nair G, Reich DS. Multiple sclerosis shrinks intralesional, and enlarges extralesional, brain parenchymal veins. Neurology 2013;80:145–151.
Sinnecker T, Dörr J, Pfueller CF, et al. Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology 2012;79:08–714.
Sinnecker T, Mittelstaedt P, Dörr J, et al. Multiple sclerosis lesions and irreversible brain tissue damage: a comparative ultrahigh-field strength magnetic resonance imaging study. Arch Neurol 2012;69:739–745.
Wuerfel J, Sinnecker T, Ringelstein EB, et al. Lesion morphology at 7 Tesla MRI differentiates Susac syndrome from multiple sclerosis. Mult Scler 2012;18:1592–1599.
Morten Blaabjerg, Klemens Ruprecht, Tim Sinnecker, Daniel Kondziella, Thoralf Niendorf, Bjørg Morell Kerrn-Jespersen, Mette Lindelof, Hans Lassmann, Bjarne Winther Kristensen, Friedemann Paul, Zsolt Illes. Widespread inflammation in CLIPPERS syndrome indicated by autopsy and ultra-high-field 7T MRI. Neurol Neuroimmunol Neuroinflamm June 2016 vol. 3 no. 3 e226